Carbodiimide
Carbodiimide is a class of organic compounds characterized by the presence of the functional group N=C=N. This group consists of a carbon atom double-bonded to two nitrogen atoms. Carbodiimides are widely used in organic synthesis and materials science for the formation of amide bonds, making them valuable reagents in the production of pharmaceuticals, polymers, and other chemical products.
Chemistry[edit]
Carbodiimides are formed through the dehydration of urea or from thiourea. The general chemical formula for carbodiimides is R-N=C=N-R', where R and R' can be the same or different alkyl or aryl groups. They are reactive towards nucleophiles, including water, alcohols, and amines, making them useful for a variety of chemical reactions.
Synthesis[edit]
The synthesis of carbodiimides typically involves the dehydration of urea derivatives or the reaction of phosgene with primary amines. One of the most common methods for preparing carbodiimides is the dehydration of urea or thiourea in the presence of a dehydrating agent, such as phosphorus oxychloride.
Reactivity[edit]
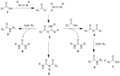
Carbodiimides are best known for their role in the formation of amide bonds from carboxylic acids and amines, a process known as amidation. This reaction is crucial in peptide synthesis, where carbodiimides, such as dicyclohexylcarbodiimide (DCC) and diisopropylcarbodiimide (DIC), are used to couple amino acids. They can also be used to activate phosphate groups in the synthesis of nucleotides and DNA.
Applications[edit]
Carbodiimides find applications in various fields, including:
- Pharmaceuticals: In drug synthesis, especially in peptide coupling reactions.
- Materials Science: In the preparation of polymers and coatings, where they are used to cross-link polymers.
- Bioconjugation: For attaching biomolecules to one another or to solid supports, crucial in the development of biosensors and diagnostic tools.
Safety and Handling[edit]
Carbodiimides are generally handled with care due to their reactivity. They can cause skin and respiratory irritation, and their reactions can release toxic by-products. Proper safety measures, including the use of personal protective equipment (PPE) and working in a fume hood, are recommended when handling these compounds.
Environmental Impact[edit]
The environmental impact of carbodiimides depends on their specific applications and disposal methods. As with many chemical reagents, responsible handling, usage, and disposal practices are essential to minimize any negative environmental effects.
Carbodiimide[edit]
-
General structure of carbodiimides
-
Crystal structure of a carbodiimide compound
-
Carbodiimide-mediated amide coupling scheme
-
Mechanism of carbodiimide reaction
-
Structure of DCC (Dicyclohexylcarbodiimide)
-
Structure of DIC (Diisopropylcarbodiimide)
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian