Tosyl group
Chemical group derived from toluenesulfonic acid
Tosyl group[edit]
The tosyl group is a functional group in organic chemistry derived from p-toluenesulfonic acid. It is often abbreviated as Ts and is represented by the chemical formula _SO2C6H4CH3. The tosyl group is commonly used as a protecting group for alcohols and amines in organic synthesis.

Structure and properties[edit]
The tosyl group consists of a sulfonyl group (_SO2_) attached to a phenyl group with a methyl group (_CH3) at the para position. This structure imparts certain chemical properties, such as the ability to act as a good leaving group in nucleophilic substitution reactions. The presence of the sulfonyl group makes the tosyl group electron-withdrawing, which stabilizes the negative charge developed during the transition state of such reactions.
Applications in organic synthesis[edit]
Tosylates, the esters of tosyl groups, are widely used in organic synthesis. They are typically formed by the reaction of an alcohol with tosyl chloride in the presence of a base. The resulting tosylate can then undergo various substitution reactions, where the tosyl group acts as a leaving group.

Protecting group[edit]
In synthetic chemistry, the tosyl group is often used to protect alcohols and amines. The protection is achieved by converting the alcohol or amine into a tosylate, which can withstand various reaction conditions. The tosyl group can later be removed by reductive or hydrolytic methods to regenerate the original functional group.

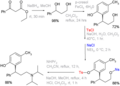
Example: Tolterodine synthesis[edit]
An example of the use of tosyl groups in synthesis is the preparation of tolterodine, a medication used to treat urinary incontinence. In one of the steps, a tosylate intermediate is formed, which is then used in further reactions to build the final product.
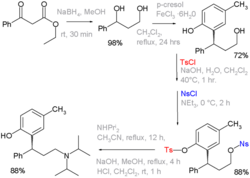
Deprotection[edit]
The removal of the tosyl group, or deprotection, can be achieved through various methods. Commonly, the tosyl group is removed by treatment with strong acids or bases, or by using reducing agents such as lithium aluminum hydride.

Related pages[edit]
References[edit]
Tosyl_group[edit]
-
Tosyl group structural formula
-
Tosylate structure
-
Tolterodine synthesis
-
Ts group
-
Ts deprotection
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian