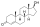Ethynylandrostanediol
| Ethynylandrostanediol | |
|---|---|

| |
| INN | |
| Drug class | |
| Routes of administration | |
| Pregnancy category | |
| Bioavailability | |
| Metabolism | |
| Elimination half-life | |
| Excretion | |
| Legal status | |
| CAS Number | 183387-50-0 |
| PubChem | 42611099 |
| DrugBank | |
| ChemSpider | 32698509 |
| KEGG | |
Ethynylandrostanediol (developmental code name HE-3235), or 17α-ethynyl-5α-androstane-3α,17β-diol, also known as Apoptone, is a synthetic, orally active, 17α-substituted analogue of the androstanediols which was under development for the treatment of prostate cancer and breast cancer but was discontinued.<ref name="pmid20814732">,
17α-alkynyl 3α, 17β-androstanediol non-clinical and clinical pharmacology, pharmacokinetics and metabolism, Invest New Drugs, 2012, Vol. 30(Issue: 1), pp. 59–78, DOI: 10.1007/s10637-010-9517-0, PMID: 20814732,</ref><ref name="pmid19337256">, Inhibition of androstenediol-dependent LNCaP tumour growth by 17alpha-ethynyl-5alpha-androstane-3alpha, 17beta-diol (HE3235), Br. J. Cancer, 2009, Vol. 100(Issue: 7), pp. 1068–72, DOI: 10.1038/sj.bjc.6604987, PMID: 19337256, PMC: 2669987,</ref><ref name="AdisInsight">http://webcache.googleusercontent.com/search?q=cache:Jy1DYIHEUtQJ:adisinsight.springer.com/drugs/800025305</ref> It has very low affinity for steroid receptors, including the AR
, ERα , ERβ , PR , and GR , and its mechanism of action is not well-characterized.<ref name="pmid20814732" /><ref name="pmid19881957">,
HE3235 inhibits growth of castration-resistant prostate cancer, Neoplasia, 2009, Vol. 11(Issue: 11), pp. 1216–25, DOI: 10.1593/neo.09960, PMID: 19881957, PMC: 2767223,</ref> It produces 5α-dihydroethisterone as an active metabolite, which may contribute importantly to its biological activity.<ref name="pmid20814732" />
References
External links
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian