Tellurium tetrafluoride
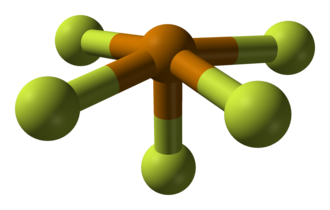
Tellurium tetrafluoride (TeF4) is a chemical compound consisting of one tellurium atom and four fluorine atoms. It is a colorless, volatile, and highly reactive solid that belongs to the group of tellurium halides. Tellurium tetrafluoride is used in chemical synthesis and has been studied for its unique properties and potential applications in materials science and nanotechnology.
Properties[edit]
Tellurium tetrafluoride is known for its distinctive properties, including its crystalline structure and reactivity. It adopts a monoclinic crystal structure at room temperature. TeF4 is soluble in solvents such as chloroform and alcohols, but it reacts with water, releasing hydrogen fluoride (HF) and forming telluric acid (H6TeO6).
Synthesis[edit]
TeF4 can be synthesized by the reaction of tellurium with fluorine gas at elevated temperatures. The reaction is highly exothermic and must be conducted under controlled conditions to prevent the formation of unwanted by-products.
Reactivity[edit]
Tellurium tetrafluoride reacts with water to form telluric acid and hydrogen fluoride. It also reacts with alkali metals and alkaline earth metals to form their respective tellurium fluorides. Its reactivity with organic compounds has been explored for organic synthesis applications.
Applications[edit]
While the direct applications of TeF4 are limited due to its reactivity and toxicity, its unique properties have made it a subject of interest in research. It has potential applications in the development of new materials, including semiconductors and nanomaterials. Additionally, its reactivity has been exploited in synthetic chemistry for the fluorination of organic compounds.
Safety[edit]
Tellurium tetrafluoride is a highly reactive and toxic compound. It poses risks of fluoride poisoning if ingested or inhaled. Appropriate safety measures, including the use of personal protective equipment (PPE) and working in a well-ventilated area or fume hood, are essential when handling TeF4.
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
