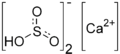Calcium bisulfite: Difference between revisions
CSV import |
No edit summary |
||
| Line 29: | Line 29: | ||
[[Category:Photographic chemicals]] | [[Category:Photographic chemicals]] | ||
[[Category:Wine terminology]] | [[Category:Wine terminology]] | ||
<gallery> | <gallery> | ||
File:Calcium_bisulfite.png|Calcium bisulfite | File:Calcium_bisulfite.png|Calcium bisulfite | ||
File:Calcium_bisulfite_ball-and-stick.png|Calcium bisulfite ball-and-stick model | File:Calcium_bisulfite_ball-and-stick.png|Calcium bisulfite ball-and-stick model | ||
</gallery> | </gallery> | ||
Latest revision as of 19:30, 16 March 2025
Calcium bisulfite (also known as calcium hydrogen sulfite) is a chemical compound with the chemical formula Ca(HSO3)2. It is a white crystalline solid that is soluble in water. It is commonly used in the food industry as a preservative and antioxidant.
Chemical properties[edit]
Calcium bisulfite is a salt of calcium, a metallic element, and bisulfite, an anion derived from sulfurous acid. It is a weak acid and can react with bases to form salts and water. When heated, it decomposes to form calcium sulfate, sulfur dioxide and water.
Uses[edit]
Calcium bisulfite is used in various industries due to its preservative and antioxidant properties. In the food industry, it is used to prevent oxidation and maintain the color of food products. It is also used in the pulp and paper industry as a bleaching agent.
In the wine industry, calcium bisulfite is used as a wine stabilizer and to prevent oxidation and preserve flavor. It is also used in the photographic industry in the development of photographic film.
Health effects[edit]
Exposure to calcium bisulfite can cause irritation to the eyes, skin, and respiratory tract. Ingestion can cause gastrointestinal disturbances. Long-term exposure can lead to more serious health effects such as damage to the respiratory system and kidneys.
See also[edit]
References[edit]
<references />
-
Calcium bisulfite
-
Calcium bisulfite ball-and-stick model