Salicylanilide: Difference between revisions
CSV import |
CSV import |
||
| Line 30: | Line 30: | ||
[[Category:Chemical compounds]] | [[Category:Chemical compounds]] | ||
{{Chemistry-stub}} | {{Chemistry-stub}} | ||
<gallery> | |||
File:Salicylanilide.svg|Salicylanilide | |||
File:Bromochlorosalicylanilide..svg|Bromochlorosalicylanilide | |||
File:Niclosamide..svg|Niclosamide | |||
File:Oxyclozanide..svg|Oxyclozanide | |||
File:Rafoxanide..svg|Rafoxanide | |||
</gallery> | |||
Revision as of 05:11, 3 March 2025
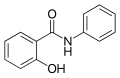
Salicylanilide is an organic compound that belongs to the class of compounds known as anilides. Anilides are derivatives of aniline, in which the hydrogen atom of the amino group has been replaced by an acyl group. Salicylanilide itself is derived from the reaction of salicylic acid and aniline, featuring both a phenol and an amide functional group. This compound is of interest in the field of medicinal chemistry due to its role as a core structure in the synthesis of various pharmaceuticals and biologically active compounds.
Chemistry
Salicylanilide is characterized by its chemical formula C_{13}H_{11}NO_{2}, indicating the presence of thirteen carbon atoms, eleven hydrogen atoms, one nitrogen atom, and two oxygen atoms. The compound's structure consists of a benzene ring (aniline part) linked to another benzene ring that contains a hydroxyl group (salicylic acid part) through an amide bond. This configuration imparts salicylanilide with unique chemical properties, such as its ability to engage in hydrogen bonding and its moderate solubility in organic solvents.
Synthesis
The synthesis of salicylanilide typically involves the acylation of aniline with salicyloyl chloride in the presence of a base, such as pyridine. This reaction proceeds via nucleophilic acyl substitution, where the amino group of aniline attacks the carbonyl carbon of salicyloyl chloride, leading to the formation of salicylanilide and the release of hydrochloric acid.
Applications
Salicylanilide and its derivatives have found applications in various fields, including:
- Medicine: Some derivatives of salicylanilide exhibit antimicrobial, antifungal, and antiparasitic properties, making them useful in the development of pharmaceuticals for treating infections.
- Agriculture: Certain salicylanilide derivatives are used as pesticides and herbicides, owing to their ability to inhibit the growth of unwanted plants and pests.
- Industry: Salicylanilide can also serve as a chemical intermediate in the synthesis of dyes, resins, and other industrial chemicals.
Safety and Toxicology
The safety profile of salicylanilide and its derivatives varies depending on the specific compound and its application. Some derivatives may be toxic to humans and animals if ingested or absorbed through the skin. Therefore, handling of salicylanilide compounds should be done with appropriate safety precautions, including the use of personal protective equipment (PPE) and adherence to regulatory guidelines.
Regulation
The use of salicylanilide derivatives in pharmaceuticals and agricultural products is subject to regulation by various governmental agencies, such as the Food and Drug Administration (FDA) in the United States and the European Medicines Agency (EMA) in the European Union. These agencies evaluate the safety, efficacy, and environmental impact of salicylanilide compounds before granting approval for their use.
See Also
-
Salicylanilide
-
Bromochlorosalicylanilide
-
Niclosamide
-
Oxyclozanide
-
Rafoxanide