Saccharin: Difference between revisions
CSV import |
CSV import |
||
| Line 1: | Line 1: | ||
'''Saccharin''' is an | == Saccharin == | ||
[[File:Saccharin.svg|thumb|right|Chemical structure of saccharin]] | |||
'''Saccharin''' is an artificial sweetener with effectively no food energy. It is about 300–400 times as sweet as [[sucrose]], but has a bitter or metallic aftertaste, especially at high concentrations. Saccharin is used to sweeten products such as drinks, candies, cookies, and medicines. | |||
== History == | == History == | ||
== | [[File:Saccharine 300 times sweeter than sugar ad, Schulze-Berge, Koechl & Movius sole licenses in the U. S. A., selling agents wanted for the Pacific Coast - Pacific wine and spirit review (IA pacificwinespiri29sanfrich) (page 10 crop).jpg|thumb|left|Advertisement for saccharin, highlighting its sweetness compared to sugar]] | ||
Saccharin was discovered in 1879 by [[Constantin Fahlberg]], a chemist working on coal tar derivatives in the laboratory of [[Ira Remsen]] at [[Johns Hopkins University]]. Fahlberg noticed a sweet taste on his hand and traced it back to the compound he had been working with. He and Remsen published their findings, but Fahlberg later patented the substance without Remsen's involvement. | |||
== Chemical Properties == | |||
[[File:Saccharin-from-xtal-3D-balls.png|thumb|right|3D ball model of saccharin]] | |||
Saccharin is a [[sulfonamide]] and a [[benzoic acid]] derivative. Its chemical formula is C<sub>7</sub>H<sub>5</sub>NO<sub>3</sub>S. It is a white, crystalline powder that is stable under heat and moderately soluble in water. | |||
== Production == | |||
[[File:Remsen-Fahlberg synthesis of saccharin.png|thumb|left|Remsen-Fahlberg synthesis of saccharin]] | |||
Saccharin can be produced by several methods. The original method, known as the Remsen-Fahlberg process, involves the reaction of [[toluene]] with [[chlorosulfonic acid]] to produce ortho-sulfobenzoic acid, which is then converted to saccharin. | |||
[[File:Maumee synthesis of saccharin.png|thumb|right|Maumee synthesis of saccharin]] | |||
Another method, the Maumee process, involves the reaction of [[phthalic anhydride]] with [[ammonia]] to form phthalimide, which is then converted to saccharin. | |||
== Uses == | |||
Saccharin is used as a non-nutritive sweetener in a variety of food and beverage products. It is often used in combination with other sweeteners to mask its metallic aftertaste. Saccharin is also used in [[toothpaste]], [[mouthwash]], and [[pharmaceuticals]]. | |||
== Health and Safety == | |||
[[File:Saccharin-Na substance photo.jpg|thumb|left|Sodium saccharin]] | |||
Saccharin has been the subject of controversy regarding its safety. Early studies in the 1970s suggested a link between saccharin and [[bladder cancer]] in laboratory rats, leading to a warning label requirement in the United States. However, subsequent research has shown that these findings are not applicable to humans, and the warning label requirement was removed in 2000. | |||
== Cultural Impact == | |||
[[File:Süßstoff Saccharin Zucker-Museum.jpg|thumb|right|Saccharin exhibit at the Sugar Museum]] | |||
Saccharin has played a significant role in the development of artificial sweeteners and has been a subject of public interest and debate. It was especially popular during [[World War I]] and [[World War II]] when sugar was rationed. | |||
Saccharin | |||
== Related Pages == | |||
* [[Aspartame]] | * [[Aspartame]] | ||
* [[Sucralose]] | * [[Sucralose]] | ||
* [[Artificial sweetener]] | |||
* [[Sugar substitute]] | |||
[[Category: | [[Category:Artificial sweeteners]] | ||
[[Category:Food additives]] | [[Category:Food additives]] | ||
[[Category: | [[Category:Sulfonamides]] | ||
Latest revision as of 14:19, 21 February 2025
Saccharin[edit]
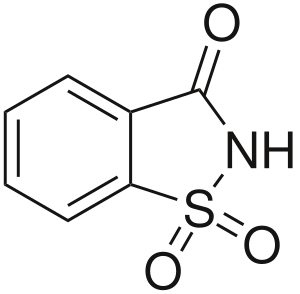
Saccharin is an artificial sweetener with effectively no food energy. It is about 300–400 times as sweet as sucrose, but has a bitter or metallic aftertaste, especially at high concentrations. Saccharin is used to sweeten products such as drinks, candies, cookies, and medicines.
History[edit]

Saccharin was discovered in 1879 by Constantin Fahlberg, a chemist working on coal tar derivatives in the laboratory of Ira Remsen at Johns Hopkins University. Fahlberg noticed a sweet taste on his hand and traced it back to the compound he had been working with. He and Remsen published their findings, but Fahlberg later patented the substance without Remsen's involvement.
Chemical Properties[edit]

Saccharin is a sulfonamide and a benzoic acid derivative. Its chemical formula is C7H5NO3S. It is a white, crystalline powder that is stable under heat and moderately soluble in water.
Production[edit]

Saccharin can be produced by several methods. The original method, known as the Remsen-Fahlberg process, involves the reaction of toluene with chlorosulfonic acid to produce ortho-sulfobenzoic acid, which is then converted to saccharin.

Another method, the Maumee process, involves the reaction of phthalic anhydride with ammonia to form phthalimide, which is then converted to saccharin.
Uses[edit]
Saccharin is used as a non-nutritive sweetener in a variety of food and beverage products. It is often used in combination with other sweeteners to mask its metallic aftertaste. Saccharin is also used in toothpaste, mouthwash, and pharmaceuticals.
Health and Safety[edit]

Saccharin has been the subject of controversy regarding its safety. Early studies in the 1970s suggested a link between saccharin and bladder cancer in laboratory rats, leading to a warning label requirement in the United States. However, subsequent research has shown that these findings are not applicable to humans, and the warning label requirement was removed in 2000.
Cultural Impact[edit]

Saccharin has played a significant role in the development of artificial sweeteners and has been a subject of public interest and debate. It was especially popular during World War I and World War II when sugar was rationed.