Alkyl group: Difference between revisions
CSV import |
CSV import |
||
| Line 9: | Line 9: | ||
File:Tert-Butyl-Skeletal-SVG.svg|Tert-butyl group | File:Tert-Butyl-Skeletal-SVG.svg|Tert-butyl group | ||
</gallery> | </gallery> | ||
== Alkyl Group == | |||
An '''alkyl group''' is a type of [[hydrocarbon]] group that is derived from an [[alkane]] by removing one [[hydrogen]] atom. Alkyl groups are often represented by the symbol R in [[organic chemistry]] and are fundamental components in the structure of many organic molecules. | |||
== Structure == | |||
Alkyl groups are composed entirely of [[carbon]] and [[hydrogen]] atoms, arranged in a chain. The simplest alkyl group is the [[methyl group]], with the formula CH__. Alkyl groups can be linear, branched, or cyclic, depending on the arrangement of the carbon atoms. | |||
=== Linear Alkyl Groups === | |||
Linear alkyl groups have a straight chain of carbon atoms. Examples include: | |||
* Methyl (CH__) | |||
* Ethyl (C_H__) | |||
* Propyl (C_H__) | |||
=== Branched Alkyl Groups === | |||
Branched alkyl groups have a carbon chain that branches off from the main chain. Examples include: | |||
* Isopropyl ((CH_)_CH_) | |||
* Sec-butyl (CH_CH_CH(CH_)_) | |||
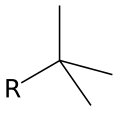
* Tert-butyl ((CH_)_C_) | |||
=== Cyclic Alkyl Groups === | |||
Cyclic alkyl groups form a ring structure. These are often referred to as [[cycloalkyl]] groups. An example is the cyclopropyl group (C_H__). | |||
== Nomenclature == | |||
The naming of alkyl groups follows the [[IUPAC nomenclature]] system. The name of an alkyl group is derived from the name of the parent alkane by replacing the "-ane" suffix with "-yl." For example, the alkane methane becomes the methyl group when one hydrogen is removed. | |||
== Properties == | |||
Alkyl groups are nonpolar and hydrophobic, meaning they do not dissolve well in water. They are typically less reactive than other functional groups, but they can participate in reactions such as [[free radical]] substitution and [[oxidation]]. | |||
== Role in Organic Chemistry == | |||
Alkyl groups are ubiquitous in organic chemistry and are found in a wide variety of compounds, including [[alcohols]], [[amines]], [[alkyl halides]], and [[esters]]. They influence the physical and chemical properties of these compounds, such as boiling point, solubility, and reactivity. | |||
== Related Pages == | |||
* [[Alkane]] | |||
* [[Functional group]] | |||
* [[Hydrocarbon]] | |||
* [[IUPAC nomenclature]] | |||
{{Organic chemistry}} | |||
[[Category:Organic chemistry]] | |||
[[Category:Functional groups]] | |||
Latest revision as of 00:36, 19 February 2025
-
Isopropyl group
-
Methyl group
-
3-Methylpentane
-
2,3,3-Trimethylpentane
-
n-Butyl group
-
Isobutyl group
-
Sec-butyl group
-
Tert-butyl group
Alkyl Group[edit]
An alkyl group is a type of hydrocarbon group that is derived from an alkane by removing one hydrogen atom. Alkyl groups are often represented by the symbol R in organic chemistry and are fundamental components in the structure of many organic molecules.
Structure[edit]
Alkyl groups are composed entirely of carbon and hydrogen atoms, arranged in a chain. The simplest alkyl group is the methyl group, with the formula CH__. Alkyl groups can be linear, branched, or cyclic, depending on the arrangement of the carbon atoms.
Linear Alkyl Groups[edit]
Linear alkyl groups have a straight chain of carbon atoms. Examples include:
- Methyl (CH__)
- Ethyl (C_H__)
- Propyl (C_H__)
Branched Alkyl Groups[edit]
Branched alkyl groups have a carbon chain that branches off from the main chain. Examples include:
- Isopropyl ((CH_)_CH_)
- Sec-butyl (CH_CH_CH(CH_)_)
- Tert-butyl ((CH_)_C_)
Cyclic Alkyl Groups[edit]
Cyclic alkyl groups form a ring structure. These are often referred to as cycloalkyl groups. An example is the cyclopropyl group (C_H__).
Nomenclature[edit]
The naming of alkyl groups follows the IUPAC nomenclature system. The name of an alkyl group is derived from the name of the parent alkane by replacing the "-ane" suffix with "-yl." For example, the alkane methane becomes the methyl group when one hydrogen is removed.
Properties[edit]
Alkyl groups are nonpolar and hydrophobic, meaning they do not dissolve well in water. They are typically less reactive than other functional groups, but they can participate in reactions such as free radical substitution and oxidation.
Role in Organic Chemistry[edit]
Alkyl groups are ubiquitous in organic chemistry and are found in a wide variety of compounds, including alcohols, amines, alkyl halides, and esters. They influence the physical and chemical properties of these compounds, such as boiling point, solubility, and reactivity.
Related Pages[edit]
| Concepts in organic chemistry |
|---|
|
|