Isotachophoresis: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 30: | Line 30: | ||
{{chemistry-stub}} | {{chemistry-stub}} | ||
{{medicine-stub}} | {{medicine-stub}} | ||
<gallery> | |||
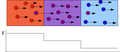
File:ITP separation model.png|ITP separation model | |||
File:Self-sharpening effect.png|Self-sharpening effect | |||
</gallery> | |||
Latest revision as of 06:14, 3 March 2025
Isotachophoresis[edit]
Isotachophoresis (ITP) is a technique used in the field of analytical chemistry and biochemistry to separate charged particles. It is based on the principle of differential migration rates of ions under an electric field. The technique is particularly useful for the separation and analysis of complex mixtures of similar compounds, such as amino acids, proteins, and nucleic acids.
Principle[edit]
The principle of isotachophoresis involves the use of two different electrolyte solutions: a leading electrolyte (LE) and a terminating electrolyte (TE). The LE has a higher mobility than the ions to be separated, while the TE has a lower mobility. When an electric field is applied, the ions in the sample migrate at different rates based on their charge and size, leading to their separation.
Procedure[edit]
The procedure for isotachophoresis begins with the preparation of the sample and the electrolyte solutions. The sample is placed between the LE and TE in a capillary tube or a gel. An electric field is then applied, causing the ions to migrate at different rates. The ions are separated into distinct zones based on their mobility, with the fastest moving ions forming a band at the front and the slowest moving ions forming a band at the back. The separated ions can then be detected and analyzed using various techniques, such as UV-Vis spectroscopy, mass spectrometry, or fluorescence spectroscopy.
Applications[edit]
Isotachophoresis has a wide range of applications in the fields of analytical chemistry and biochemistry. It is used for the separation and analysis of complex mixtures of similar compounds, such as amino acids, proteins, and nucleic acids. It is also used in the detection of drugs and their metabolites in biological samples, in the analysis of environmental samples for pollutants, and in the study of ion transport in biological systems.
See Also[edit]
References[edit]
<references />
-
ITP separation model
-
Self-sharpening effect