Glyoxylic acid: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 26: | Line 26: | ||
{{Chem-stub}} | {{Chem-stub}} | ||
{{food-stub}} | {{food-stub}} | ||
<gallery> | |||
File:Glyoxylic_acid_hydration.png|Glyoxylic acid hydration | |||
File:Glyoxylic_acid_hydrate_dimerization.png|Glyoxylic acid hydrate dimerization | |||
File:Glyoxylic_acid_H-bonded.png|Glyoxylic acid H-bonded | |||
File:Glyoxylate_metabolism_in_hepatocytes.jpg|Glyoxylate metabolism in hepatocytes | |||
File:Photorespiration_in_arabidopsis.jpg|Photorespiration in Arabidopsis | |||
</gallery> | |||
Latest revision as of 04:37, 18 February 2025
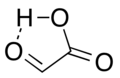
Glyoxylic acid or oxoacetic acid is an organic compound that is both an aldehyde and a carboxylic acid. Glyoxylic acid is a colorless solid that occurs naturally and can be prepared in the laboratory. It is the smallest alpha-keto acid.
Structure and properties[edit]
Glyoxylic acid contains both aldehyde and carboxylic acid functional groups. The molecular formula for glyoxylic acid is C2H2O3. It is a colorless solid that is highly reactive and readily forms hydrates.
Occurrence and preparation[edit]
Glyoxylic acid occurs naturally in various plants, fruits, and vegetables. It can also be prepared in the laboratory by oxidizing glycolic acid, oxalic acid, or acetic acid with nitric acid.
Uses[edit]
Glyoxylic acid is used in various industries. It is used in the production of pharmaceuticals, herbicides, and dyes. It is also used in the food industry as a flavoring agent and in the cosmetic industry for hair straightening treatments.
Safety[edit]
Glyoxylic acid is a strong acid and can cause burns if it comes into contact with the skin or eyes. It is also harmful if swallowed or inhaled.
See also[edit]
References[edit]
<references />
-
Glyoxylic acid hydration
-
Glyoxylic acid hydrate dimerization
-
Glyoxylic acid H-bonded
-
Glyoxylate metabolism in hepatocytes
-
Photorespiration in Arabidopsis