Tuinal
Tuinal[edit]
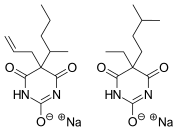

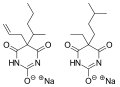
Tuinal is a combination barbiturate medication that contains equal parts of two barbiturate salts: secobarbital and amobarbital. It was introduced in the late 1940s by Eli Lilly and Company as a sedative and hypnotic for the treatment of insomnia and other sleep disorders.
History[edit]
Tuinal was first introduced in 1946 and became widely used in the 1950s and 1960s. It was marketed as a prescription medication for patients who required a strong sedative. However, due to its potential for abuse and addiction, its use declined over the years.
Pharmacology[edit]
Tuinal acts as a central nervous system depressant. The combination of secobarbital and amobarbital enhances the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) in the brain, leading to sedative and hypnotic effects. The drug has a rapid onset of action and a relatively short duration of effect.
Medical Uses[edit]
Tuinal was primarily prescribed for the short-term treatment of severe insomnia. It was also used as a pre-anesthetic agent and for the treatment of certain types of seizures. Due to its high potential for dependence and overdose, it is no longer commonly prescribed.
Abuse and Dependence[edit]
Tuinal has a high potential for abuse and can lead to physical and psychological dependence. Overdose can result in severe respiratory depression, coma, and death. As a result, its use is now highly restricted.
Legal Status[edit]
Due to its potential for abuse, Tuinal is classified as a Schedule II controlled substance in the United States. This classification indicates that it has a high potential for abuse, with use potentially leading to severe psychological or physical dependence.
Related Pages[edit]
References[edit]
-
Chemical structure of Secobarbital and Amobarbital
-
Tuinal capsules
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian