Oxygen fluoride
Chemical compounds of oxygen and fluorine
Oxygen fluorides are a group of compounds consisting of oxygen and fluorine. These compounds are notable for their reactivity and are used in various chemical applications. The most common oxygen fluorides are oxygen difluoride (OF2), dioxygen difluoride (O2F2), and trioxygen difluoride (O3F2).
Oxygen Difluoride[edit]
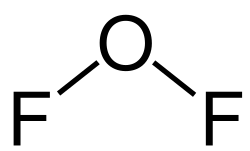
Oxygen difluoride (OF2) is a chemical compound with a bent molecular geometry. It is a pale yellow gas at room temperature and is known for its strong oxidizing properties. Oxygen difluoride is used in various industrial applications, including as an oxidizer in rocket propellants.
Properties[edit]
Oxygen difluoride is a highly reactive compound. It reacts with water to form hydrogen fluoride and oxygen. The compound is also known to react with many metals and nonmetals, often forming fluorides and oxides.
Preparation[edit]
Oxygen difluoride can be prepared by the reaction of fluorine with a dilute solution of sodium hydroxide:
- 2 F2 + 2 NaOH → OF2 + 2 NaF + H2O
Dioxygen Difluoride[edit]
Dioxygen difluoride (O2F2) is a compound with a very unusual structure and extreme reactivity. It is a reddish-orange solid at low temperatures and decomposes explosively at temperatures above −160 °C.
Properties[edit]
Dioxygen difluoride is known for its ability to oxidize almost any material, including noble gases like xenon. It is one of the most powerful oxidizing agents known.
Preparation[edit]
Dioxygen difluoride is typically prepared by the reaction of fluorine with oxygen at low temperatures:
- O2 + F2 → O2F2
Trioxygen Difluoride[edit]
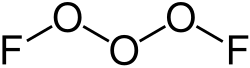
Trioxygen difluoride (O3F2) is a less common oxygen fluoride. It is a powerful oxidizing agent and is used in specialized chemical syntheses.
Properties[edit]
Trioxygen difluoride is a highly reactive compound, capable of oxidizing a wide range of substances. It is less stable than oxygen difluoride and is typically handled with care in controlled environments.
Preparation[edit]
The preparation of trioxygen difluoride involves the reaction of ozone with fluorine under controlled conditions:
- O3 + F2 → O3F2
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian