Trioxsalen
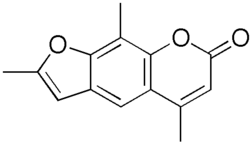
Trioxsalen is a furocoumarin and a psoralen derivative. It is used in combination with ultraviolet light in the treatment of vitiligo and psoriasis, a process known as PUVA therapy.
Chemistry[edit]
Trioxsalen is a synthetic tricyclic compound and is one of several furocoumarin derivatives used in PUVA therapy. It is structurally related to psoralen, but contains three oxygen atoms (hence the prefix 'trioxs') instead of two.
Mechanism of Action[edit]
Trioxsalen works by intercalating into the DNA of skin cells, forming covalent bonds with the DNA upon exposure to ultra violet (UV) light. This process, known as photoaddition, results in the formation of monoadducts and crosslinks that can inhibit DNA replication and transcription, leading to cell death. This is particularly effective in rapidly dividing cells, such as those found in psoriatic plaques.
Uses[edit]
Trioxsalen is used in PUVA therapy, a treatment for skin conditions such as psoriasis, vitiligo, and eczema. The patient takes trioxsalen orally or applies it topically, then exposes the affected skin to UVA light. This causes the skin cells to die off, reducing the symptoms of these conditions.
Side Effects[edit]
Common side effects of trioxsalen include nausea, headache, and itching. More serious side effects can include severe burns from the UVA light and an increased risk of skin cancer.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
