Triosephosphate isomerase
Enzyme involved in glycolysis and gluconeogenesis
Triosephosphate isomerase (TPI or TIM) is an enzyme that plays a critical role in the glycolysis and gluconeogenesis pathways. It catalyzes the reversible interconversion of the three-carbon sugars dihydroxyacetone phosphate (DHAP) and glyceraldehyde 3-phosphate (G3P).
Structure[edit]
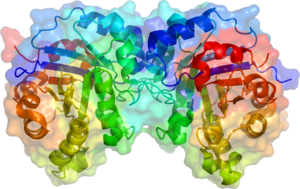
Triosephosphate isomerase is a dimeric enzyme, with each monomer consisting of about 250 amino acids. The enzyme is characterized by an eight-stranded α/β barrel, a common protein fold known as the TIM barrel. This structure is highly conserved across different species, indicating its evolutionary importance.
Mechanism[edit]
The enzyme catalyzes the isomerization of DHAP to G3P through an enediol intermediate. The reaction involves the abstraction of a proton from the substrate by a glutamate residue in the active site, followed by the transfer of a hydrogen atom to form the product. This reaction is essential for efficient energy production in cells.
Function[edit]
Triosephosphate isomerase is crucial for the efficient functioning of glycolysis and gluconeogenesis. By converting DHAP to G3P, it ensures that the energy yield from glucose metabolism is maximized. This conversion is necessary because only G3P can continue through the subsequent steps of glycolysis to produce ATP.
Thermodynamics[edit]
The reaction catalyzed by triosephosphate isomerase is near equilibrium under physiological conditions, with a small change in free energy. This allows the reaction to proceed readily in both directions, depending on the cellular needs for energy production or glucose synthesis.
Clinical significance[edit]
Deficiency in triosephosphate isomerase activity can lead to a rare genetic disorder known as triosephosphate isomerase deficiency, which is characterized by hemolytic anemia and neurological dysfunction. This highlights the enzyme's importance in normal cellular metabolism.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian