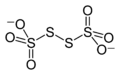
Thiosulfate
Thiosulfate is an oxyanion of sulfur. The chemical formula for thiosulfate is S2O3^2-. It is a compound that contains sulfur in two different oxidation states: +2 and +6. Thiosulfates are salts or esters of thiosulfuric acid, and they are commonly found in a variety of chemical and biological systems. They play a significant role in several biochemical processes, including detoxification pathways and in the treatment of cyanide poisoning.
Chemistry[edit]
Thiosulfate is characterized by its tetrahedral geometry, where a central sulfur atom is bonded to three oxygen atoms and one sulfur atom through a single bond. This unique structure contributes to its reactivity and its ability to act as a reducing agent. Thiosulfates are stable in neutral or alkaline solutions but decompose in acidic conditions to produce sulfur dioxide, sulfur, and water.
Production[edit]
Thiosulfates can be produced through several methods. One common laboratory method involves the reaction of sulfur dioxide with aqueous solutions of sulfur, which yields thiosulfate. Industrially, thiosulfates are often produced as by-products in the manufacture of sodium sulfide or during the leaching of metal sulfide ores in the presence of oxygen.
Applications[edit]
Photography[edit]
In photography, thiosulfates, particularly sodium thiosulfate (Na2S2O3), are used as fixing agents. They work by dissolving the unreacted silver halides from the photographic emulsion, making the image permanent and insensitive to light.
Medical[edit]
In the medical field, sodium thiosulfate is used in the treatment of cyanide poisoning. It acts by converting toxic cyanide ions into thiocyanate, which is less harmful and can be excreted by the kidneys. Additionally, it has been investigated for its potential in treating calciphylaxis in patients with end-stage renal disease.
Industrial[edit]
Thiosulfates are also used in various industrial applications, including water treatment, where they serve as oxygen scavengers to prevent corrosion in water distribution systems. They are also used in the leather tanning industry and as components in certain types of cement.
Environmental Impact[edit]
Thiosulfates are considered relatively benign from an environmental perspective. However, their decomposition products, such as sulfur dioxide, can contribute to air pollution and acid rain if released in large quantities.
Safety[edit]
Thiosulfates are generally regarded as safe when handled properly. However, exposure to large amounts can cause irritation to the skin, eyes, and respiratory system. Appropriate safety measures, such as wearing protective equipment, should be taken when handling chemicals containing thiosulfate.
See Also[edit]
-
Thiosulfate ion 2D dimensions
-
Tetrathionate ion 2D
-
Thiosulfate
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian