Tetrasodium EDTA
Tetrasodium EDTA[edit]
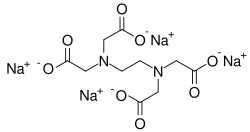
Tetrasodium EDTA is a sodium salt of ethylenediaminetetraacetic acid (EDTA), a chelating agent that is widely used in various industrial and medical applications. It is a white, water-soluble solid that is commonly used to sequester metal ions in aqueous solutions.
Chemical Properties[edit]
Tetrasodium EDTA has the chemical formula C__H__N_Na_O_ and a molar mass of 380.17 g/mol. It is a derivative of EDTA, which is a polyamino carboxylic acid. The tetrasodium form is the sodium salt, which makes it more soluble in water compared to the acid form.
Applications[edit]
Tetrasodium EDTA is used in a variety of applications due to its ability to bind metal ions. Some of the key applications include:
Industrial Applications[edit]
- Water Treatment: It is used to soften water by sequestering calcium and magnesium ions, which are responsible for water hardness.
- Detergents and Cleaners: Tetrasodium EDTA is added to detergents to enhance their cleaning efficiency by binding metal ions that could otherwise interfere with the cleaning process.
Medical Applications[edit]
- Pharmaceuticals: It is used as a preservative and stabilizer in various pharmaceutical formulations.
- Cosmetics: In the cosmetic industry, tetrasodium EDTA is used to improve the stability and appearance of products by preventing metal ion contamination.
Safety and Environmental Impact[edit]
Tetrasodium EDTA is generally considered safe for use in the concentrations typically found in consumer products. However, it can have environmental impacts if released in large quantities, as it can bind to metal ions in the environment and affect aquatic life.
Related Compounds[edit]
See Also[edit]
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian