Stereospecificity
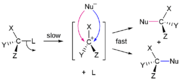
File:StereospecificCarbeneAddition.svg
Stereospecificity is a property of a chemical reaction in which the mechanism of the reaction leads to the preferential formation of a specific stereoisomer when multiple stereoisomers are possible. This concept is crucial in the field of stereochemistry, which studies the spatial arrangement of atoms in molecules and how this affects their chemical behavior.
Overview[edit]
Stereospecificity is distinct from stereoselectivity, although the two terms are often confused. While stereoselectivity refers to the preference for the formation of one stereoisomer over another, stereospecificity implies that the reaction mechanism inherently leads to the formation of a specific stereoisomer. This means that the starting materials' stereochemistry directly influences the stereochemistry of the product.
Types of Stereospecific Reactions[edit]
Stereospecific reactions can be classified into several types based on the nature of the stereoisomers involved:
- Enantiospecific reactions: These reactions produce a specific enantiomer from a chiral starting material.
- Diastereospecific reactions: These reactions produce a specific diastereomer from a chiral or achiral starting material.
Examples[edit]
One classic example of a stereospecific reaction is the SN2 reaction. In an SN2 reaction, the nucleophile attacks the carbon atom from the opposite side of the leaving group, leading to an inversion of configuration at the carbon center. This reaction is stereospecific because the stereochemistry of the product is directly determined by the stereochemistry of the starting material.
Another example is the Diels-Alder reaction, which is a cycloaddition reaction between a diene and a dienophile. The stereochemistry of the diene and dienophile directly influences the stereochemistry of the product, making the reaction stereospecific.
Applications[edit]
Stereospecificity is particularly important in the field of pharmaceutical chemistry, where the biological activity of a drug can be highly dependent on its stereochemistry. Many drugs are chiral, and often only one enantiomer is therapeutically active. Therefore, stereospecific synthesis is crucial for the production of effective and safe pharmaceuticals.
See Also[edit]
- Stereochemistry
- Chirality (chemistry)
- Enantiomer
- Diastereomer
- SN2 reaction
- Diels-Alder reaction
- Pharmaceutical chemistry
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
