Sodium gluconate
A chemical compound used in various applications
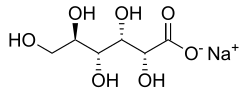
Sodium gluconate is the sodium salt of gluconic acid. It is a white, granular, crystalline solid, very soluble in water. Sodium gluconate is widely used in various industries due to its excellent chelating properties, particularly in alkaline and concentrated alkaline solutions.
Chemical properties[edit]
Sodium gluconate has the chemical formula C_H__NaO_. It is a compound that is stable in the presence of oxidizing agents and is non-corrosive, non-toxic, and readily biodegradable. The compound is known for its ability to chelate metal ions, which makes it useful in a variety of applications.
Applications[edit]
Construction industry[edit]
In the construction industry, sodium gluconate is used as a set retarder and plasticizer in concrete admixtures. It helps to delay the setting time of concrete, allowing for more time to work with the material before it hardens.
Food industry[edit]
Sodium gluconate is used as a food additive and is recognized for its ability to enhance flavor and preserve food. It is often used in the formulation of processed foods and beverages.
Pharmaceuticals[edit]
In the pharmaceutical industry, sodium gluconate is used as a stabilizer for medicines and as a component in oral care products. Its non-toxic nature makes it suitable for use in products that are ingested or applied to the body.
Cleaning products[edit]
Sodium gluconate is a common ingredient in cleaning products due to its ability to soften water and enhance the cleaning power of detergents. It is effective in removing mineral deposits and scale from surfaces.
Biodegradability[edit]
One of the significant advantages of sodium gluconate is its biodegradability. It breaks down into harmless substances in the environment, making it an environmentally friendly choice for various applications.
Safety[edit]
Sodium gluconate is considered safe for use in food and pharmaceutical products. It is non-toxic and does not pose significant health risks when used appropriately.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian