Sodium–potassium alloy
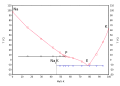
Sodium–potassium alloy, often referred to as NaK (pronounced nack), is an alloy of two alkali metals, sodium (Na) and potassium (K), and it is usually liquid at room temperature. Various compositions of NaK can exist, but the most common mixture contains 40% sodium and 60% potassium, which is eutectic in nature, meaning it has a lower melting point than either of its two components. NaK is highly reactive with water and air, necessitating careful handling and specific storage conditions, typically under an inert gas atmosphere such as argon or nitrogen.
Properties[edit]
NaK alloys are characterized by their low density, high thermal conductivity, and excellent heat transfer properties. These characteristics make NaK useful as a coolant in various applications, including nuclear reactors, where its ability to remain liquid at room temperature and conduct heat efficiently is highly valued. However, the reactivity of NaK with water and air, producing hydrogen gas and potassium hydroxide, poses significant risks, requiring rigorous safety protocols.
Applications[edit]
The primary use of NaK alloys is in the cooling systems of nuclear reactors, where their excellent thermal properties help manage reactor temperatures efficiently. NaK is also used in some chemical synthesis processes as a strong reducing agent and in certain types of aerospace equipment for heat transfer purposes.
Safety[edit]
Handling NaK requires strict safety measures due to its reactivity. Exposure to air can lead to oxidation, while contact with water results in a vigorous reaction that produces hydrogen gas and heat, potentially leading to explosions. Therefore, NaK is usually stored and handled under an inert gas atmosphere in well-ventilated areas. Personal protective equipment (PPE) is mandatory when working with NaK to prevent skin and eye contact.
Environmental Impact[edit]
The environmental impact of NaK is primarily associated with its potential to release hydrogen gas upon reaction with water, posing a risk of explosion and fire. Proper disposal methods are critical to minimize any environmental risks, including neutralization with alcohol before disposal.
See Also[edit]
-
NaK Alloy
-
Phase diagram potassium sodium solid liquid
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian