Semagacestat
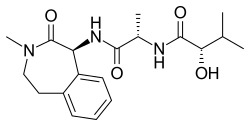
Semagacestat is an experimental drug that was investigated for the treatment of Alzheimer's disease. It functions as a gamma secretase inhibitor, which was hypothesized to reduce the production of beta-amyloid peptides, a key component in the pathogenesis of Alzheimer's disease. Despite initial promise, the development of Semagacestat was discontinued due to concerns over its efficacy and safety profile.
Development and Clinical Trials[edit]
The development of Semagacestat was part of a broader effort to address Alzheimer's disease by targeting the amyloid cascade hypothesis. This hypothesis suggests that the accumulation of beta-amyloid peptides in the brain is a critical factor in the disease's progression. By inhibiting gamma secretase, an enzyme involved in the production of beta-amyloid, researchers hoped to slow or halt the progression of Alzheimer's disease.
Clinical trials for Semagacestat, however, revealed significant challenges. In Phase III trials, not only did the drug fail to demonstrate a slowing of cognitive decline in patients with Alzheimer's, but it also was associated with worsening of cognitive function compared to placebo. Additionally, there were concerns about adverse effects, including an increased risk of skin cancer and infections.
Mechanism of Action[edit]
Semagacestat inhibits the activity of gamma secretase, a complex enzyme that plays a crucial role in the production of beta-amyloid peptides. Gamma secretase cleaves the amyloid precursor protein (APP) into smaller fragments, including beta-amyloid. By inhibiting this process, Semagacestat aimed to reduce the overall production of beta-amyloid peptides.
Discontinuation[edit]
The development of Semagacestat was discontinued following the negative outcomes of the Phase III clinical trials. The results raised significant concerns about the viability of gamma secretase inhibitors as a treatment strategy for Alzheimer's disease. The failure of Semagacestat and similar compounds led to increased scrutiny of the amyloid cascade hypothesis and prompted researchers to explore alternative therapeutic targets and strategies.
Impact on Alzheimer's Research[edit]
The discontinuation of Semagacestat had a profound impact on the field of Alzheimer's research. It prompted a reevaluation of the amyloid cascade hypothesis and the strategies employed to target Alzheimer's disease. The failure of Semagacestat and other similar drugs underscored the complexity of Alzheimer's disease and the need for a more nuanced understanding of its pathogenesis. It also highlighted the importance of identifying biomarkers for early detection and intervention, as well as the potential for targeting multiple pathways involved in the disease process.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian