Relebactam
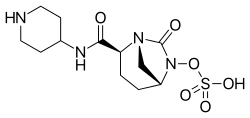
Relebactam is a beta-lactamase inhibitor used in combination with antibiotics to treat certain bacterial infections. It is specifically designed to inhibit the action of beta-lactamase enzymes produced by bacteria, which can degrade beta-lactam antibiotics and render them ineffective.
Mechanism of Action[edit]
Relebactam works by binding to the active site of beta-lactamase enzymes, thereby preventing these enzymes from breaking down beta-lactam antibiotics. This inhibition allows the antibiotics to retain their antibacterial activity and effectively kill or inhibit the growth of bacteria.
Clinical Use[edit]
Relebactam is commonly used in combination with imipenem, a broad-spectrum beta-lactam antibiotic. The combination of relebactam and imipenem is marketed under the brand name Recarbrio. This combination is used to treat complicated urinary tract infections (cUTIs), complicated intra-abdominal infections (cIAIs), and hospital-acquired bacterial pneumonia (HABP) and ventilator-associated bacterial pneumonia (VABP).
Pharmacokinetics[edit]
Relebactam is administered intravenously and has a half-life that allows for dosing every six hours when combined with imipenem. It is primarily excreted unchanged in the urine.
Side Effects[edit]
Common side effects of relebactam, when used in combination with imipenem, include nausea, diarrhea, headache, and infusion site reactions. Serious side effects may include allergic reactions and seizures, particularly in patients with a history of central nervous system disorders.
Development and Approval[edit]
Relebactam was developed by Merck & Co. and received approval from the United States Food and Drug Administration (FDA) in 2019 for use in combination with imipenem.
See Also[edit]
- Beta-lactam antibiotic
- Beta-lactamase
- Imipenem
- Antibiotic resistance
- Complicated urinary tract infection
- Complicated intra-abdominal infection
- Hospital-acquired bacterial pneumonia
- Ventilator-associated bacterial pneumonia
References[edit]
External Links[edit]
| Relebactam
| |
|---|---|
| Trade names | Recarbrio
|
| Routes | Intravenous
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian