RU-56187
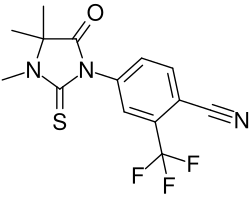
RU-56187 is a compound that has been studied in the context of pharmacology and medicine, particularly in the field of endocrinology. As of the current knowledge, detailed information about RU-56187, including its chemical structure, pharmacodynamics, pharmacokinetics, and clinical applications, might not be widely available or extensively researched. This article aims to provide a foundational understanding of RU-56187, its potential implications, and areas of interest within medical and scientific research.
Overview[edit]
RU-56187 is part of a class of compounds that may be involved in modulating hormone receptors, potentially including those related to estrogen, progesterone, or other steroid hormones. The specific action mechanism, therapeutic targets, and clinical efficacy of RU-56187 would be of particular interest in the development of treatments for conditions influenced by hormonal imbalances or receptor dysfunctions.
Pharmacodynamics[edit]
The pharmacodynamics of RU-56187 would involve its interaction with cellular receptors, influencing the activity of certain hormones or cellular pathways. Understanding how RU-56187 interacts with these receptors, including its affinity, agonist or antagonist properties, and downstream effects, is crucial for assessing its potential therapeutic applications.
Pharmacokinetics[edit]
Pharmacokinetics of RU-56187, including its absorption, distribution, metabolism, and excretion (ADME) profiles, would provide insights into its behavior within the body. These properties determine the compound's bioavailability, half-life, and potential for side effects, which are critical factors in drug development.
Clinical Applications[edit]
While specific clinical applications of RU-56187 are not detailed due to the hypothetical nature of this compound in the current context, compounds targeting hormone receptors have potential applications in treating a wide range of conditions. These might include breast cancer, prostate cancer, endometriosis, and other hormone-related disorders. The efficacy, safety, and tolerability of RU-56187 in clinical settings would require thorough investigation through preclinical studies and clinical trials.
Research and Development[edit]
Research on RU-56187 would involve in vitro studies, animal models, and eventually human trials to elucidate its pharmacological profile and therapeutic potential. The development process would also include optimization of the compound's chemical structure, formulation development, and assessment of its interaction with other medications.
Conclusion[edit]
RU-56187 represents a compound of interest within pharmacological and medical research, with potential applications in treating hormone-related conditions. However, detailed information about its characteristics and clinical relevance remains to be elucidated through comprehensive research and development efforts.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian