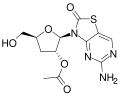
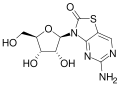
RG7795
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these messages)
|
| RG7795
| |
|---|---|
| Legal status | Investigational |
| CAS Number | 1174920-78-5
|
RG7795 (previously ANA773) is an antiviral drug candidate that as of 2015 had been in Phase II trials in hepatitis B.[1] It is an orally-available prodrug of isatoribine,[2] that was under development by Anadys Pharmaceuticals when it was acquired by Roche in 2011.[3] Its active metabolite is an [agonist]] of TLR7; activation of TLR7 causes secretion of endogenous type 1 interferons, which have antiviral activity.[2]
References[edit]
- ↑ RG 7795(link). AdisInsight.
- ↑ 2.0 2.1 "Tickling the TLR7 to cure viral hepatitis.".Journal of Translational Medicine.14 May 2014;12
- 129.doi:10.1186/1479-5876-12-129.PMID:24884741.PMC:4039542.
- ↑ , Inovio Goes It Alone on Hepatitis B Immunotherapy Vaccine as Roche Ends Collaboration Full text, Genetic Engineering News, August 3, 2016,
RG7795[edit]
-
RG-7795
-
Isatoribine
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian