Protiofate
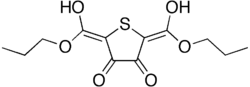
Protiofate is a chemical compound that belongs to the class of organic compounds known as alkanes. It is a type of hydrocarbon that contains only single bonds. Protiofate is a stable, non-reactive substance due to the absence of double or triple bonds. It is used in various industrial applications due to its stability and non-reactivity.
Chemical Structure[edit]
The chemical structure of Protiofate consists of a chain of carbon atoms, each bonded to hydrogen atoms. The number of carbon atoms can vary, leading to different forms of Protiofate. The general formula for Protiofate is CnH2n+2, where n is the number of carbon atoms.
Properties[edit]
Protiofate is a colorless, odorless gas at room temperature. It is insoluble in water but soluble in organic solvents such as ethanol and acetone. It has a low boiling point and a high melting point. Protiofate is non-polar, which means it does not have a positive or negative charge and does not readily react with other substances.
Uses[edit]
Protiofate is used in a variety of industrial applications. It is used as a fuel in internal combustion engines due to its high energy content. It is also used as a raw material in the production of other chemicals, such as plastics and synthetic rubber. In addition, Protiofate is used in the manufacture of pharmaceuticals and cosmetics.
Safety[edit]
Protiofate is generally considered safe to handle, but it can be harmful if inhaled in large quantities. It can also cause skin and eye irritation. Therefore, it is important to use appropriate safety measures when handling Protiofate.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
