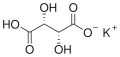
Potassium bitartrate
Potassium bitartrate, also known as cream of tartar, is a byproduct of winemaking. It is an acid salt that has a number of uses in cooking and other fields.
Chemical properties[edit]
Potassium bitartrate (chemical formula KC4H5O6) is a white, crystalline powder. It is a byproduct of the fermentation process in winemaking, where it crystallizes on the inside of wine barrels. The crystals are then collected and purified to produce the commercial product.
Potassium bitartrate is an acid salt, which means it is the product of a reaction between an acid and a base. In this case, the acid is tartaric acid and the base is potassium hydroxide.
Uses[edit]
Potassium bitartrate has a number of uses in cooking, particularly in baking. It is used as a leavening agent in some recipes, where it reacts with baking soda to produce carbon dioxide gas and help dough rise. It can also be used to stabilize egg whites and prevent sugar from crystallizing in candy making.
In addition to its culinary uses, potassium bitartrate has applications in other fields. For example, it is used in the production of potassium hydrogen tartrate, which is used in the manufacture of mirrors. It is also used in the production of tartar emetic, a compound used in the treatment of certain medical conditions.
Safety[edit]
Potassium bitartrate is generally considered safe for consumption. However, like all substances, it can be harmful if consumed in large amounts. Symptoms of potassium bitartrate overdose can include nausea, vomiting, diarrhea, and abdominal pain.
See also[edit]
-
Structural formula of Potassium bitartrate
-
Crystals of Potassium bitartrate
-
Commercial cream of tartar containing Potassium bitartrate
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian