Potassium carbonate
Overview[edit]
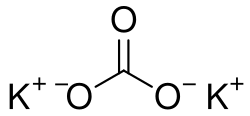
Potassium carbonate is a white salt, soluble in water, which forms a strongly alkaline solution. It is deliquescent, often appearing a damp or wet solid. Potassium carbonate is used in the production of soap and glass, as well as in the food industry.
Chemical Properties[edit]
Potassium carbonate has the chemical formula K2CO3. It is an inorganic compound that is a white, water-soluble salt. It is often used in the laboratory as a mild drying agent where other drying agents such as calcium chloride and magnesium sulfate may be incompatible.
Production[edit]
Potassium carbonate is produced by the reaction of potassium hydroxide with carbon dioxide:
- 2 KOH + CO2 _ K2CO3 + H2O
This reaction is similar to the production of sodium carbonate (soda ash) from sodium hydroxide.
Uses[edit]
Potassium carbonate is used in various applications:
- Glass Manufacturing: It is used in the production of specialty glass, such as optical glass.
- Soap Production: Historically, it was used in the production of soap by saponification of fats.
- Food Industry: It is used as a food additive, identified as E501, and is used as a buffering agent.
- Laboratory Use: It is used as a drying agent for some chemical reactions.
Safety[edit]
Potassium carbonate is generally considered safe when handled properly. However, it is a strong alkaline substance and can cause irritation to the skin and eyes. Proper safety precautions, such as wearing gloves and goggles, should be taken when handling this chemical.
Related Compounds[edit]
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian