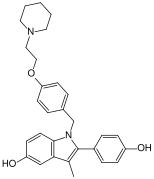
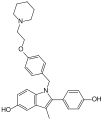
Pipendoxifene
A selective estrogen receptor modulator
| Pipendoxifene | |
|---|---|
|
| |
| IUPAC name | 1-[4-[(1Z)-2-(4-hydroxyphenyl)-3-(4-piperidin-1-ylbutoxy)phenyl]phenyl]ethanone
|
Pipendoxifene is a selective estrogen receptor modulator (SERM) that has been studied for its potential use in the treatment of breast cancer and other estrogen-related conditions. It is known for its ability to act as an estrogen receptor antagonist in breast tissue while potentially acting as an agonist in other tissues such as bone.
Mechanism of Action[edit]
Pipendoxifene functions by binding to estrogen receptors in various tissues. As a SERM, it exhibits tissue-selective activities, meaning it can block or activate estrogen receptors depending on the tissue type. In breast tissue, pipendoxifene acts as an antagonist, inhibiting the proliferative action of estrogen, which is beneficial in the treatment of estrogen receptor-positive breast cancer. In bone tissue, it may act as an agonist, helping to maintain bone density and reduce the risk of osteoporosis.
Pharmacokinetics[edit]
The pharmacokinetic profile of pipendoxifene involves its absorption, distribution, metabolism, and excretion. It is typically administered orally, and its bioavailability can be influenced by factors such as food intake and individual metabolic differences. The drug is metabolized primarily in the liver and excreted through the kidneys.
Clinical Applications[edit]
Pipendoxifene has been investigated for its potential use in the treatment of breast cancer, particularly in postmenopausal women. Its ability to selectively modulate estrogen receptors makes it a candidate for reducing the risk of breast cancer recurrence. Additionally, its effects on bone density suggest potential use in the prevention of osteoporosis in postmenopausal women.
Side Effects[edit]
As with other SERMs, pipendoxifene may have side effects, including hot flashes, leg cramps, and an increased risk of venous thromboembolism. The risk-benefit profile must be carefully considered in patients with a history of thromboembolic events.
Research and Development[edit]
Research on pipendoxifene is ongoing, with studies focusing on its efficacy and safety in various populations. Clinical trials are essential to determine its long-term effects and potential as a therapeutic agent in estrogen-related conditions.
Related pages[edit]
-
Pipendoxifene
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian