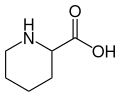
Pipecolic acid
Pipecolic acid, also known as piperidine-2-carboxylic acid, is an organic compound and a non-proteinogenic alpha amino acid. Structurally, it is related to the amino acid proline, with the main difference being the presence of an extra methylene group in its ring structure, making it a six-membered ring compared to proline's five-membered ring. Pipecolic acid is found in many living organisms and plays a role in various biochemical processes. It is particularly notable for its occurrence in lysine degradation in humans and other mammals.
Structure and Properties[edit]
Pipecolic acid is a cyclic amino acid, with the chemical formula C_6H_11NO_2. Its structure consists of a six-membered piperidine ring, with a carboxylic acid (-COOH) and an amino (-NH_2) group attached to the second carbon atom of the ring. This compound exists in two enantiomeric forms, L-Pipecolic acid and D-Pipecolic acid, due to the presence of a chiral center at the carbon bearing the amino group. L-Pipecolic acid is the biologically active isomer found in nature.
Biosynthesis and Metabolism[edit]
Pipecolic acid is synthesized in living organisms through two main pathways: the lysine degradation pathway and the direct hydroxylation of lysine. In humans and other mammals, the predominant pathway for pipecolic acid production is through the degradation of lysine in the liver, specifically through the saccharopine pathway. This process involves several enzymatic steps that convert lysine into pipecolic acid, which can then be further metabolized or participate in other biochemical pathways.
Biological Role[edit]
In humans, pipecolic acid has been implicated in several physiological and pathological processes. It is found in higher concentrations in the brain and has been associated with the regulation of neurotransmitter release, suggesting a role in the central nervous system. Elevated levels of pipecolic acid have been observed in patients with peroxisomal disorders, such as Zellweger syndrome, and in those with defects in lysine degradation, indicating its potential as a biomarker for these conditions.
Clinical Significance[edit]
The accumulation of pipecolic acid in bodily fluids is a characteristic of several inherited metabolic disorders. For instance, hyperpipecolic acidemia is a condition characterized by elevated levels of pipecolic acid in the plasma, often associated with neurological impairments and developmental delays. The measurement of pipecolic acid levels in plasma or urine can be used in the diagnosis and monitoring of these metabolic diseases.
Research[edit]
Research on pipecolic acid has focused on its role in metabolic pathways, its potential therapeutic applications, and its involvement in disease mechanisms. Studies have explored its neuroprotective effects, its role as a signaling molecule, and its potential use in drug development, particularly in the design of enzyme inhibitors and receptor modulators.
See Also[edit]
Pipecolic acid[edit]
-
Structural formula of pipecolic acid
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian