Peptide synthesis
Peptide Synthesis[edit]
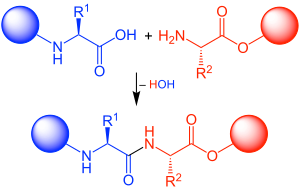
Peptide synthesis is the process of creating peptides, which are short chains of amino acids linked by peptide bonds. This process is fundamental in the field of biochemistry and molecular biology, as peptides play crucial roles in biological functions and are used in research and therapeutic applications.
Methods of Peptide Synthesis[edit]
Peptide synthesis can be performed using various methods, each with its own advantages and limitations. The two primary methods are liquid-phase peptide synthesis and solid-phase peptide synthesis.
Liquid-Phase Peptide Synthesis[edit]
Liquid-phase peptide synthesis involves the sequential addition of amino acids in solution. This method was historically significant but has largely been replaced by solid-phase techniques due to the latter's efficiency and ease of purification.
Solid-Phase Peptide Synthesis[edit]

Solid-phase peptide synthesis (SPPS) is the most common method used today. It involves anchoring the C-terminal amino acid of the peptide to an insoluble resin, allowing for the sequential addition of protected amino acids. This method was pioneered by Robert Bruce Merrifield, who was awarded the Nobel Prize in Chemistry in 1984 for this innovation.
Coupling Reagents[edit]
The formation of peptide bonds requires activation of the carboxyl group of the incoming amino acid. Various coupling reagents are used to facilitate this process.
Carbodiimide Reagents[edit]
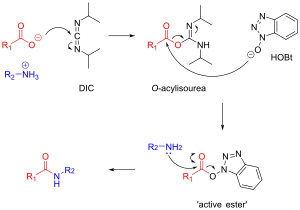
Carbodiimides, such as dicyclohexylcarbodiimide (DCC) and diisopropylcarbodiimide (DIC), are commonly used in peptide synthesis. They activate the carboxyl group to form an active ester, which then reacts with the amine group of the next amino acid.
Additives[edit]
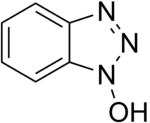
Additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt) are often used to suppress racemization and improve yields.

Protecting Groups[edit]
Protecting groups are essential in peptide synthesis to prevent unwanted side reactions. The most common protecting groups are used for the N-terminus and side chains of amino acids.
N-Terminus Protecting Groups[edit]

The carbobenzyloxy (Cbz), tert-butyloxycarbonyl (Boc), and 9-fluorenylmethyloxycarbonyl (Fmoc) groups are commonly used to protect the N-terminus of amino acids.
Side Chain Protecting Groups[edit]
Side chain protecting groups are used to protect functional groups on the amino acid side chains that might react during peptide synthesis.
Deprotection[edit]
After the peptide chain is assembled, the protecting groups must be removed to yield the final peptide.
Boc Deprotection[edit]

Boc groups are removed using strong acids such as trifluoroacetic acid (TFA).
Fmoc Deprotection[edit]

Fmoc groups are removed using mild bases such as piperidine.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian