Enantiomer
Enantiomer
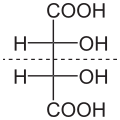
An Enantiomer is one of two stereoisomers that are mirror images of each other that are non-superposable (not identical), much as one's left and right hands are the same except for opposite orientation. This is a concept in stereochemistry, a sub-discipline of chemistry.
Overview[edit]
Enantiomers are a type of chiral molecule. Chirality is a property of a molecule that results from its structure. It is the property of a molecule that makes it non-superposable on its mirror image.
Enantiomers have the same physical properties (such as boiling point, melting point, and density), but they interact differently with plane polarized light and with other chiral molecules. This difference in behavior is due to the spatial arrangement of the atoms in the molecule.
Properties[edit]
Enantiomers have identical physical properties except for the direction in which they rotate polarized light. This rotation is measured using a polarimeter and is expressed as the optical rotation.
Applications[edit]
Enantiomers are of great importance in biochemistry, the study of chemical processes in living organisms, and in the pharmaceutical industry. Many drugs are chiral, and the two enantiomers can have different effects on the body.
See Also[edit]
References[edit]
-
Lactic acid enantiomers
-
Meso-tartaric acid mirror image
-
Mecoprop enantiomers
-
Citalopram structural formula
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian