Nucleophilic conjugate addition
Nucleophilic conjugate addition is a type of organic reaction and a key concept in the field of organic chemistry. It involves the addition of a nucleophile to a conjugated system, typically an alkene or alkyne, that has an electron-withdrawing group attached to it. This process is crucial for the formation of many important compounds in both nature and synthetic chemistry.
Mechanism[edit]
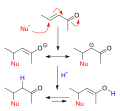
The mechanism of nucleophilic conjugate addition involves the nucleophile attacking the β-carbon of a conjugated system, which is the carbon atom adjacent to the carbon atom bearing the electron-withdrawing group. This attack leads to the formation of a new chemical bond between the nucleophile and the β-carbon, resulting in the addition of the nucleophile to the conjugated system.
The electron-withdrawing group plays a crucial role in this reaction by stabilizing the negative charge that is formed on the β-carbon during the reaction. This stabilization is typically achieved through resonance or inductive effects, making the β-carbon more susceptible to nucleophilic attack.
Types of Nucleophiles[edit]
Nucleophiles that participate in conjugate addition reactions can be varied and include:
- Water (H₂O)
- Alcohols
- Amines
- Thiols
- Organometallic compounds such as Grignard reagents and organolithium reagents
Applications[edit]
Nucleophilic conjugate addition is widely used in the synthesis of various organic compounds. It is particularly useful in the construction of carbon-carbon bonds and the introduction of functional groups into molecules. Applications include:
- Synthesis of pharmaceuticals and agrochemicals
- Preparation of natural products
- Construction of complex molecular architectures in material science
Examples[edit]
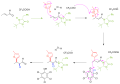
One of the most well-known examples of nucleophilic conjugate addition is the Michael addition, where a nucleophile adds to an α,β-unsaturated carbonyl compound. Another example is the addition of organometallic compounds to α,β-unsaturated nitriles or esters.
See Also[edit]
References[edit]
-
Nucleophilic conjugate addition
-
Example of conjugate addition
-
(R)-3-phenyl-cyclohexanone
-
Enantioselective organo cascade catalysis
-
Conjugated addition of thiols
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian