Nitrous acid
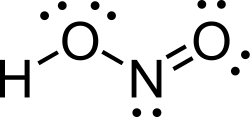
Nitrous acid is a weak and monobasic acid known by the chemical formula HNO₂. It is not typically found in a pure form but is usually prepared in situ by the acidification of aqueous solutions of its salts, the nitrites. Nitrous acid is an important intermediate in the nitrogen cycle and plays a significant role in various chemical reactions, including the formation of diazonium compounds and the decomposition of nitrites.
Structure[edit]
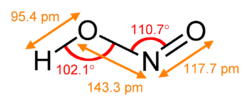
Nitrous acid exists in two isomeric forms: the more stable trans form and the less stable cis form. The trans form is characterized by the hydrogen atom and the oxygen atom being on opposite sides of the nitrogen atom, while in the cis form, they are on the same side.
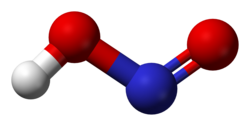
The molecular geometry of nitrous acid is bent, with the nitrogen atom bonded to one oxygen atom via a double bond and to another oxygen atom via a single bond, which also carries the hydrogen atom. This configuration results in a polar molecule with a significant dipole moment.
Preparation[edit]
Nitrous acid is typically prepared by the reaction of a nitrite salt with a mineral acid, such as hydrochloric acid:
- \[ \text{NaNO}_2 + \text{HCl} \rightarrow \text{HNO}_2 + \text{NaCl} \]
This reaction is usually carried out in an aqueous solution, as nitrous acid is unstable and decomposes rapidly in the presence of water.
Reactions[edit]
Nitrous acid is involved in several important chemical reactions:
- Diazotization: It reacts with amines to form diazonium salts, which are key intermediates in the synthesis of azo dyes and other aromatic compounds.
- Decomposition: In aqueous solutions, nitrous acid decomposes to form nitric oxide (NO) and nitrogen dioxide (NO₂):
- \[ 2 \text{HNO}_2 \rightarrow \text{NO}_2 + \text{NO} + \text{H}_2\text{O} \]
- Oxidation and Reduction: Nitrous acid can act as both an oxidizing and a reducing agent, depending on the reaction conditions and the reactants involved.
Role in the Environment[edit]
Nitrous acid plays a crucial role in the atmosphere as a source of hydroxyl radicals (OH), which are important for the degradation of pollutants. It is also involved in the formation of photochemical smog and can influence the ozone balance in the lower atmosphere.