NS-304
NS-304[edit]
NS-304, also known as Selexipag, is a pharmaceutical drug used in the treatment of pulmonary arterial hypertension (PAH). It is a selective prostacyclin receptor agonist that helps to dilate blood vessels and reduce blood pressure in the lungs, thereby improving symptoms and exercise capacity in patients with PAH.
Mechanism of Action[edit]
NS-304 acts as an agonist of the prostacyclin receptor (IP receptor). Prostacyclin is a naturally occurring molecule in the body that has vasodilatory and antiproliferative effects on the pulmonary artery smooth muscle cells. By activating the IP receptor, NS-304 mimics the effects of prostacyclin, leading to vasodilation and inhibition of smooth muscle cell proliferation, which are beneficial in managing PAH.
Pharmacokinetics[edit]
NS-304 is administered orally and is metabolized in the liver to its active metabolite, ACT-333679. The drug has a half-life of approximately 8 hours, allowing for twice-daily dosing. It is primarily excreted in the urine.
Clinical Use[edit]
NS-304 is indicated for the treatment of pulmonary arterial hypertension (PAH) to delay disease progression and improve exercise capacity. It is often used in combination with other PAH therapies, such as endothelin receptor antagonists or phosphodiesterase-5 inhibitors.
Side Effects[edit]
Common side effects of NS-304 include headache, diarrhea, nausea, and jaw pain. These side effects are generally mild to moderate in severity. Patients should be monitored for signs of liver dysfunction and other adverse effects.
Clinical Trials[edit]
The efficacy and safety of NS-304 were established in the GRIPHON trial, a large, multicenter, randomized, placebo-controlled study. The trial demonstrated that NS-304 significantly reduced the risk of morbidity and mortality in patients with PAH.
Also see[edit]
- Pulmonary arterial hypertension
- Prostacyclin
- Endothelin receptor antagonist
- Phosphodiesterase-5 inhibitor
| NS-304 | |
|---|---|
|
| |
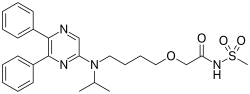
| IUPAC name | 2-[4-[(5,6-diphenylpyrazin-2-yl)(isopropyl)amino]butoxy]-N-(methylsulfonyl)acetamide
|
| Routes of administration | Oral
|
| Metabolism | Hepatic |
| Elimination half-life | 8 hours |
| Excretion | Urine
|
| CAS Number | 475086-01-2 |
| PubChem | 11949646 |
| DrugBank | DB09277 |
| ChemSpider | 10128256 |
| KEGG | D08910
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian