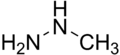Monomethylhydrazine
Monomethylhydrazine (also known as MMH or 1,1-Dimethylhydrazine) is a volatile, colorless liquid that is used as a rocket propellant and a precursor to pharmaceuticals. It is derived from hydrazine, a simple compound that consists of two nitrogen atoms and two hydrogen atoms.
Chemical Properties[edit]
Monomethylhydrazine is a derivative of hydrazine, with one of the hydrogen atoms replaced by a methyl group. It has the chemical formula CH3(NH)NH2. It is a volatile, colorless liquid that is miscible with water and most organic solvents. It has a strong, fishy odor and is highly toxic.
Uses[edit]
Monomethylhydrazine is primarily used as a rocket propellant in bipropellant rocket engines because it is hypergolic with various oxidizers such as nitrogen tetroxide (N2O4) and nitric acid (HNO3). It is also used in the manufacture of pharmaceuticals and agricultural chemicals.
Health Effects[edit]
Exposure to monomethylhydrazine can cause irritation to the eyes, skin, and respiratory tract. It can also cause headaches, nausea, and dizziness. Long-term exposure can lead to liver and kidney damage. It is also a suspected carcinogen.
Safety[edit]
Due to its high reactivity and toxicity, monomethylhydrazine requires careful handling. It should be stored in a cool, well-ventilated area away from sources of ignition. It should also be kept away from oxidizing agents and acids to prevent violent reactions.
See Also[edit]
Monomethylhydrazine[edit]
-
Monomethylhydrazine
-
Methylhydrazine 3D balls
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian