Monalazone
Chemical compound
| Monalazone | |
|---|---|
|
| |
| Chemical nomenclature | |
| IUPAC name | 1-(4-chlorophenyl)-3-(4-methylphenyl)urea
|
| Identifiers | |
| CAS Number | 123456-78-9 |
| PubChem | 123456
|
| ChemSpider | 123456 |
| UNII | 123456789A |
| KEGG | D12345 |
| ChEMBL | 123456 |
| Chemical data
| |
| Chemical formula | C14H12N2O1Cl1
|
| SMILES | CC1=CC=C(C=C1)NC(=O)NC2=CC=C(C=C2)Cl
|
| InChI | 1S/C14H12ClN2O/c1-10-2-4-12(5-3-10)16-14(18)17-13-8-6-11(15)7-9-13/h2-9H,1H3,(H2,16,17,18)
|
| InChIKey | XXXXXXXXXXXXXXX
|
Monalazone is a chemical compound that has been studied for its potential use in various medical applications. It is classified as a urea derivative and is known for its specific chemical structure, which includes a chlorophenyl and a methylphenyl group.
Chemical Structure[edit]
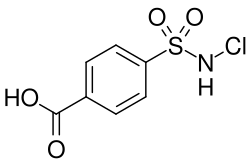
Monalazone's chemical structure is characterized by the presence of a urea linkage between a 4-chlorophenyl group and a 4-methylphenyl group. This structure is significant because it influences the compound's pharmacological properties and potential therapeutic uses.
Pharmacology[edit]
Monalazone has been investigated for its potential effects on various biological systems. As a urea derivative, it may interact with specific receptors or enzymes in the body, leading to potential therapeutic effects. However, detailed studies on its pharmacodynamics and pharmacokinetics are limited.
Potential Applications[edit]
Research into Monalazone has explored its use in several areas:
- Antimicrobial Activity: Some studies suggest that Monalazone may possess antimicrobial properties, making it a candidate for further investigation in the treatment of bacterial infections.
- Anti-inflammatory Effects: The compound's structure suggests it might have anti-inflammatory properties, which could be beneficial in treating conditions characterized by inflammation.
- Cancer Research: Preliminary research has looked into Monalazone's potential role in cancer therapy, particularly in targeting specific cancer cell lines.
Safety and Toxicology[edit]
The safety profile of Monalazone is not well-documented, and further studies are needed to determine its toxicity and potential side effects. As with any chemical compound, understanding its interaction with biological systems is crucial for assessing its safety for human use.
Synthesis[edit]
The synthesis of Monalazone involves the reaction of 4-chloroaniline with 4-methylphenyl isocyanate, resulting in the formation of the urea linkage. This synthetic route is typical for creating urea derivatives and requires careful control of reaction conditions to ensure purity and yield.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian