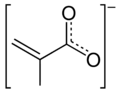
Methacrylate
Methacrylate refers to a chemical compound derived from methacrylic acid. This compound is a key component in the production of various plastic materials and resins, known for their transparency, resistance to breakage, and elasticity. Methacrylates are widely used in the medical field, dentistry, and in the manufacturing of paints, adhesives, and coatings.
Chemical Properties[edit]
Methacrylate compounds contain the methacryloyl group, which is characterized by the chemical formula C4H6O2. This group is highly reactive, making methacrylates suitable for polymerization processes. When methacrylates undergo polymerization, they form polymethyl methacrylate (PMMA), a transparent thermoplastic often used as a lightweight, shatter-resistant alternative to glass.
Applications[edit]
Medical Applications[edit]
In the medical field, methacrylate polymers are used in a variety of applications. They are a key material in the production of dental prosthetics and dental fillings, owing to their durability and compatibility with human tissue. Methacrylates are also used in the manufacturing of contact lenses and intraocular lenses, which require materials that are not only transparent but also biocompatible.
Industrial Applications[edit]
Beyond healthcare, methacrylates play a crucial role in the production of paints, adhesives, and coatings. Their ability to form strong, durable, and flexible bonds makes them ideal for use in automotive paints, industrial coatings, and sealants. Methacrylate adhesives are known for their strong bonding capabilities across a wide range of materials, including metals, plastics, and ceramics.
Environmental and Health Concerns[edit]
While methacrylates are invaluable in various industries, they are not without their environmental and health concerns. Exposure to methacrylate vapors can cause irritation to the eyes, skin, and respiratory system. In the workplace, proper ventilation and protective equipment are essential to minimize exposure risks. Environmental regulations also govern the disposal and handling of methacrylate compounds to prevent contamination of water and soil.
Regulation and Safety[edit]
Regulatory bodies, such as the Environmental Protection Agency (EPA) and the Occupational Safety and Health Administration (OSHA), have established guidelines for the safe handling, use, and disposal of methacrylate compounds. These regulations are designed to protect both human health and the environment from potential hazards associated with methacrylate use.
Conclusion[edit]
Methacrylates are versatile compounds with a wide range of applications in the medical, dental, and industrial fields. Their unique properties make them indispensable in the production of various products, from medical devices to automotive paints. However, it is crucial to adhere to safety guidelines and regulations to mitigate any potential health and environmental risks.
-
Methacrylate anion 2D skeletal
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian