MK-434
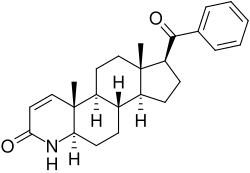
MK-434 is a compound that falls within the realm of pharmacology and medicinal chemistry. As of the current knowledge base, specific details about MK-434, including its chemical structure, pharmacological properties, and potential therapeutic applications, might not be widely documented or could be part of ongoing research within the pharmaceutical industry. In the context of drug development, compounds like MK-434 often undergo a series of stages including discovery, preclinical research, and clinical trials to determine their safety, efficacy, and potential market use.
Discovery and Development[edit]
The discovery of new pharmaceutical compounds like MK-434 typically involves the identification of potential drug targets and the synthesis of compounds that can modulate the activity of these targets. This process can be guided by computer-aided drug design, high-throughput screening, and other biotechnological methods. The development phase for MK-434 would include optimization of its chemical properties to improve its efficacy, bioavailability, and safety profile.
Pharmacological Properties[edit]
The pharmacological properties of MK-434, such as its mechanism of action, pharmacokinetics (absorption, distribution, metabolism, and excretion), and pharmacodynamics (the biochemical and physiological effects of the drug and its mechanisms of action), are crucial for understanding its potential therapeutic benefits and side effects. These properties are determined through preclinical studies in vitro (in test tubes) and in vivo (in animals).
Clinical Trials[edit]
Before MK-434 can be approved for use in humans, it must undergo rigorous clinical trials to evaluate its safety and efficacy. These trials are typically conducted in phases, starting with small studies in healthy volunteers (Phase I) to assess safety and dosing, followed by larger studies in patients to determine efficacy and further evaluate safety (Phase II), and finally, large-scale studies to confirm its efficacy, monitor side effects, compare it to commonly used treatments, and collect information that will allow the drug to be used safely (Phase III).
Potential Therapeutic Applications[edit]
The therapeutic applications of MK-434 would depend on its target and mechanism of action. For instance, if MK-434 is found to inhibit a key enzyme involved in the progression of a specific disease, it could be developed as a treatment for that condition. The potential benefits, risks, and clinical outcomes associated with its use would be closely examined through clinical research.
Regulatory Approval and Market[edit]
Following successful clinical trials, MK-434 would need to gain regulatory approval before it can be marketed and prescribed. This involves a review of all the scientific data by regulatory bodies, such as the Food and Drug Administration (FDA) in the United States, to ensure that the benefits of the drug outweigh its risks.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
