Ketose
A type of monosaccharide containing a ketone group
Ketose is a type of monosaccharide (simple sugar) that contains a ketone group. Ketoses are important in various biological processes and are found in many natural sources. Unlike aldoses, which contain an aldehyde group, ketoses have a carbonyl group (C=O) located at the second carbon atom of the sugar chain.
Structure[edit]
Ketoses are characterized by the presence of a ketone group, which is a carbon atom double-bonded to an oxygen atom (C=O). This group is typically located at the second carbon atom in the sugar chain, distinguishing ketoses from aldoses, where the carbonyl group is at the terminal carbon.
Common Ketoses[edit]
Some common ketoses include:
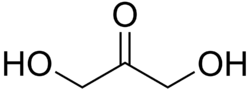
- Dihydroxyacetone - The simplest ketose, with the chemical formula C_H_O_. It is a triose, meaning it contains three carbon atoms.
- Fructose - A hexose sugar with the formula C_H__O_, commonly found in fruits and honey. It is one of the most important ketoses in human metabolism.
- Ribulose - A pentose sugar with the formula C_H__O_, involved in the Calvin cycle of photosynthesis.
Biological Importance[edit]
Ketoses play crucial roles in various metabolic pathways. For example, fructose is a key player in the glycolysis pathway, where it is phosphorylated and converted into intermediates that enter the energy-producing cycles of the cell.
Metabolism[edit]
In the human body, ketoses are metabolized through pathways such as glycolysis and the pentose phosphate pathway. Fructose, for instance, is converted into fructose-1-phosphate by the enzyme fructokinase, and then further processed into intermediates that can be used for energy production or biosynthesis.
Sources[edit]
Ketoses are naturally found in various foods. Fructose, for example, is abundant in fruits, vegetables, and honey. It is also a component of sucrose, a disaccharide composed of glucose and fructose.
Chemical Properties[edit]
Ketoses can undergo isomerization to form aldoses through a process called tautomerization. This isomerization is facilitated by the presence of an enediol intermediate, allowing the conversion between the ketone and aldehyde forms.
Images[edit]

Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian