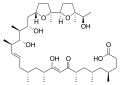
Ionomycin
Ionomycin is a polyether antibiotic that is derived from Streptomyces conglobatus. It is a divalent cation ionophore and is more selective for calcium than other divalent cations. It is used in research to raise the intracellular level of calcium and to study the effects of this on cellular processes.
Structure[edit]
Ionomycin is a complex molecule with a large number of oxygen atoms, which allow it to complex with divalent cations. It has a cyclic structure with a large number of ether groups, which contribute to its ability to transport ions across lipid membranes.
Mechanism of action[edit]
Ionomycin works by forming a complex with calcium ions, which it then transports across the lipid bilayer of cells. This increases the intracellular concentration of calcium, which can then interact with various intracellular targets. This can lead to a variety of effects, depending on the cell type and the specific processes that are sensitive to calcium levels.
Uses[edit]
Ionomycin is used in research to study the effects of increased intracellular calcium levels. This can be useful in a variety of fields, including cell biology, neuroscience, and pharmacology. It is also used in research on apoptosis, or programmed cell death, as increased intracellular calcium levels can trigger this process.
Safety[edit]
As with any substance, the safety of ionomycin depends on the dose and the specific circumstances of its use. In general, it should be handled with care to avoid accidental ingestion or skin contact. It should also be stored properly to prevent degradation.
See also[edit]
References[edit]
-
Ionomycin (free acid)
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian