Iberdomide
Immunomodulatory drug
| Iberdomide
| |
|---|---|
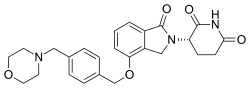
| IUPAC name | (3S)-3-(4-amino-1-oxoisoindolin-2-yl)piperidine-2,6-dione
|
| CAS Number | 1323403-33-3 |
| PubChem | 49846518
|
| ChemSpider | 32700838
|
Iberdomide is an immunomodulatory drug that is being investigated for its potential use in the treatment of various autoimmune diseases and hematologic malignancies. It is a derivative of thalidomide and belongs to a class of drugs known as cereblon (CRBN) modulators.
Mechanism of Action[edit]
Iberdomide exerts its effects by binding to the cereblon protein, which is part of the E3 ubiquitin ligase complex. This binding leads to the degradation of specific target proteins, including Ikaros (IKZF1) and Aiolos (IKZF3), which are transcription factors involved in the regulation of immune responses. By degrading these proteins, iberdomide modulates the activity of T cells and B cells, thereby exerting its immunomodulatory effects.
Clinical Development[edit]
Iberdomide is currently undergoing clinical trials for the treatment of various conditions, including multiple myeloma, systemic lupus erythematosus (SLE), and other autoimmune disorders. Early-phase clinical trials have shown promising results, with iberdomide demonstrating efficacy in reducing disease activity and improving clinical outcomes in patients with these conditions.
Side Effects[edit]
As with other immunomodulatory drugs, iberdomide may cause a range of side effects. Common side effects observed in clinical trials include fatigue, nausea, diarrhea, and cytopenias such as neutropenia and thrombocytopenia. Patients receiving iberdomide should be monitored for these potential adverse effects.
Regulatory Status[edit]
As of now, iberdomide has not yet received regulatory approval for clinical use. It remains an investigational drug, and its safety and efficacy continue to be evaluated in ongoing clinical trials.
See Also[edit]
- Thalidomide
- Lenalidomide
- Pomalidomide
- Immunomodulatory drugs
- Multiple myeloma
- Systemic lupus erythematosus
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian