Histidine methyl ester
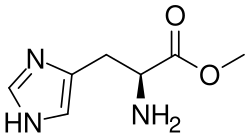
0Histidine methyl ester is a derivative of the amino acid histidine. It is formed by the methylation of the carboxyl group of histidine, resulting in an ester linkage. This modification can occur naturally in biological systems, or it can be synthetically produced in a laboratory setting.
Structure and Properties[edit]
Histidine methyl ester has a similar structure to histidine, with the exception of the methyl ester group replacing the carboxyl group. This modification results in a compound that is more lipophilic (fat-soluble) than histidine, which can influence its interactions with other molecules and its behavior in biological systems.
The methyl group in the ester linkage is a small, non-polar group, which can influence the properties of the molecule. For example, it can increase the molecule's hydrophobicity, potentially altering its solubility in various solvents and its interactions with other molecules.
Biological Role[edit]
In biological systems, the methylation of histidine to form histidine methyl ester can be catalyzed by enzymes known as methyltransferases. This modification can have various effects on the function of the histidine residue and the protein in which it is incorporated.
For example, methylation can influence the charge of the histidine residue, potentially affecting its ability to form hydrogen bonds and its interactions with other molecules. This can, in turn, influence the structure and function of the protein.
Synthetic Production[edit]
In a laboratory setting, histidine methyl ester can be synthesized through the reaction of histidine with a methylating agent, such as methyl iodide. This reaction typically requires a base to deprotonate the carboxyl group of histidine, allowing it to react with the methylating agent.
Applications[edit]
Histidine methyl ester has potential applications in various fields, including biochemistry, pharmacology, and medicine. For example, it can be used as a building block in the synthesis of peptides and proteins, or as a probe in studies of protein structure and function.
In pharmacology, histidine methyl ester and its derivatives may have potential as therapeutic agents. For example, they could be used to modulate the activity of proteins or enzymes, or to target specific cells or tissues in the body.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
