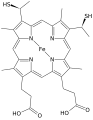
Heme C
Heme C[edit]
Heme C is a type of heme, which is an essential prosthetic group found in various hemoproteins. It plays a crucial role in biological processes such as electron transfer and catalysis. Heme C is distinct from other heme types, such as Heme A and Heme B, due to its unique covalent linkages to the protein structure.
Structure[edit]
Heme C is characterized by its iron-containing porphyrin ring, which is covalently attached to the protein via thioether bonds. These bonds are formed between the vinyl groups of the porphyrin and cysteine residues of the apoprotein. This covalent attachment is a defining feature that distinguishes heme C from other heme types, which are typically non-covalently bound to their proteins.
The porphyrin ring in heme C is known as protoporphyrin IX, which is also found in heme B. However, the covalent attachment in heme C provides additional stability and influences the redox properties of the heme.
Function[edit]
Heme C is primarily involved in electron transfer processes. It is a key component of cytochromes, particularly cytochrome c, which is a vital part of the electron transport chain in mitochondria. Cytochrome c transfers electrons between Complex III (cytochrome bc1 complex) and Complex IV (cytochrome c oxidase) in the mitochondrial inner membrane.
The redox potential of heme C is finely tuned by its protein environment, allowing it to efficiently participate in electron transfer reactions. This tuning is crucial for the proper functioning of the electron transport chain and, consequently, for ATP production through oxidative phosphorylation.
Biosynthesis[edit]
The biosynthesis of heme C involves the incorporation of iron into protoporphyrin IX to form heme B, followed by the covalent attachment of the heme to the apoprotein. This process is facilitated by specific enzymes that catalyze the formation of thioether bonds between the vinyl groups of the heme and the cysteine residues of the protein.
Clinical Significance[edit]
Alterations in heme C-containing proteins can lead to various diseases. For example, mutations in cytochrome c can result in impaired electron transport and are associated with conditions such as mitochondrial myopathies and neurodegenerative diseases. Understanding the structure and function of heme C is therefore important for developing therapeutic strategies for these conditions.
Also see[edit]
Template:Heme Template:Cytochromes
-
Heme c
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian