Guaiacolsulfonic acid
Chemical compound
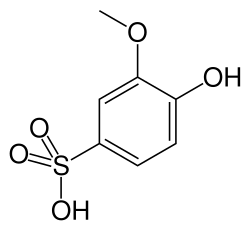
Guaiacolsulfonic acid is a chemical compound that is primarily used in the medical field as an expectorant. It is a derivative of guaiacol, which is a naturally occurring organic compound derived from guaiacum or wood creosote. Guaiacolsulfonic acid is known for its ability to help clear mucus from the airways, making it easier for patients to breathe.
Chemical Structure and Properties[edit]
Guaiacolsulfonic acid is characterized by the presence of a sulfonic acid group attached to the guaiacol molecule. The chemical formula for guaiacolsulfonic acid is C7H8O5S. The addition of the sulfonic acid group increases the solubility of the compound in water, which is beneficial for its use in medicinal formulations.

Pharmacological Action[edit]
As an expectorant, guaiacolsulfonic acid works by thinning and loosening mucus in the airways. This action helps to clear congestion and makes it easier for patients to cough up phlegm. It is often included in cough syrups and other over-the-counter medications designed to treat symptoms of colds, bronchitis, and other respiratory conditions.
Medical Uses[edit]
Guaiacolsulfonic acid is used in the treatment of respiratory conditions where mucus production is a problem. It is particularly useful in conditions such as chronic bronchitis, asthma, and chronic obstructive pulmonary disease (COPD). By facilitating the removal of mucus, it helps to improve breathing and reduce the risk of infection.
Safety and Side Effects[edit]
Guaiacolsulfonic acid is generally considered safe when used as directed. However, some patients may experience side effects such as nausea, vomiting, or gastrointestinal discomfort. It is important for patients to follow dosing instructions carefully and to consult with a healthcare provider if they experience any adverse effects.
Related Compounds[edit]
Guaiacolsulfonic acid is related to other expectorants such as guaifenesin, which is also used to treat respiratory conditions. Both compounds share a similar mechanism of action but differ in their chemical structure and solubility properties.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian