Ganirelix
Ganirelix[edit]
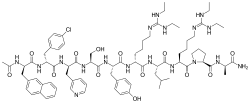
Ganirelix is a synthetic decapeptide that acts as a gonadotropin-releasing hormone antagonist (GnRH antagonist). It is primarily used in assisted reproduction to prevent premature ovulation during controlled ovarian hyperstimulation.
Mechanism of Action[edit]
Ganirelix works by competitively binding to the gonadotropin-releasing hormone receptors in the pituitary gland. This action inhibits the secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which are crucial for the maturation and release of oocytes from the ovaries. By suppressing these hormones, Ganirelix effectively prevents premature ovulation, allowing for better timing of oocyte retrieval in in vitro fertilization (IVF) procedures.
Clinical Use[edit]
Ganirelix is administered via subcutaneous injection. It is typically used in assisted reproductive technology (ART) protocols, particularly in IVF cycles. The drug is usually started in the mid-to-late follicular phase of the menstrual cycle, following the initiation of follicle-stimulating hormone (FSH) therapy.
Dosage and Administration[edit]
The standard dosage of Ganirelix is 250 micrograms per day. It is administered until the day of human chorionic gonadotropin (hCG) administration, which is used to trigger final oocyte maturation.
Side Effects[edit]
Common side effects of Ganirelix include abdominal pain, headache, and injection site reactions such as redness or swelling. Rarely, it may cause hypersensitivity reactions.
Advantages[edit]
Ganirelix offers several advantages in ART protocols, including a reduced risk of ovarian hyperstimulation syndrome (OHSS) compared to gonadotropin-releasing hormone agonist protocols. It also allows for a shorter duration of treatment and more flexible scheduling of oocyte retrieval.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian