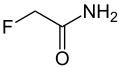
Fluoroacetamide
Fluoroacetamide is a chemical compound with the formula CH2FCONH2. It is an organofluorine compound that functions primarily as a rodenticide. Due to its high toxicity to mammals, its use is restricted in many countries, and it is considered a hazardous substance that requires careful handling.
Chemistry[edit]
Fluoroacetamide is a fluorinated analogue of acetamide. It is synthesized by the reaction of acetic acid with ammonia under specific conditions that allow for the introduction of a fluorine atom. The presence of the fluorine atom significantly alters the chemical and biological properties of the compound compared to its non-fluorinated counterpart.
Mechanism of Action[edit]
The toxic effects of fluoroacetamide are primarily due to its interference with cellular metabolism. Once ingested, it is metabolized to fluoroacetic acid, which inhibits the aconitase enzyme. This inhibition disrupts the citric acid cycle (Krebs cycle), a critical pathway for energy production in cells. The disruption leads to a severe decrease in ATP production, resulting in energy depletion and eventually cell death.
Uses[edit]
Historically, fluoroacetamide has been used as a rodenticide to control populations of rodents that pose a threat to agriculture and public health. However, its use has been significantly reduced or banned in many jurisdictions due to its high toxicity and the risk it poses to non-target species, including humans.
Toxicity[edit]
Fluoroacetamide is highly toxic to mammals, including humans. Symptoms of poisoning can include nausea, vomiting, convulsions, and cardiac distress. Due to its mode of action, there is no specific antidote for fluoroacetamide poisoning, making exposure potentially fatal. Treatment is generally supportive and aimed at managing symptoms.
Environmental Impact[edit]
The environmental impact of fluoroacetamide is a concern due to its high toxicity and persistence. It can affect non-target species, including birds, insects, and aquatic organisms. The risk of secondary poisoning, where predators are poisoned after consuming affected prey, is particularly significant.
Regulation[edit]
Due to its high toxicity and environmental risks, the use of fluoroacetamide as a rodenticide is heavily regulated or banned in many countries. Regulations typically focus on restricting its availability, controlling its use in the environment, and ensuring proper disposal of residues.
See Also[edit]
-
Fluoracetamide structural formula
-
Fluoroacetamide 3D balls
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian