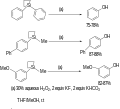
Fleming–Tamao oxidation
Fleming–Tamao Oxidation is a chemical reaction that involves the oxidation of organosilanes to alcohols using peroxides or other oxidants. This reaction is significant in organic chemistry for the selective transformation of silyl ethers into alcohols, a process that is both efficient and environmentally benign. The reaction is named after Ian Fleming and Tohru Tamao, who independently developed this methodology in the late 20th century.
Mechanism[edit]
The Fleming–Tamao oxidation proceeds through a series of steps beginning with the oxidation of the silicon center in the organosilane. This is typically achieved using hydrogen peroxide (H2O2) or tert-butyl hydroperoxide (TBHP) as the oxidant. The initial step generates a silanol, which under the reaction conditions undergoes a chemical rearrangement to form a silyl ether. Subsequent hydrolysis of the silyl ether yields the corresponding alcohol and a silanol by-product, which can be easily removed from the reaction mixture.
Applications[edit]
This reaction is widely used in the synthesis of complex organic molecules, including natural products and pharmaceuticals. Its ability to selectively oxidize silyl ethers to alcohols without affecting other functional groups makes it a valuable tool in the chemist's arsenal. The Fleming–Tamao oxidation has found applications in the synthesis of steroids, terpenes, and polyketides, among other classes of compounds.
Advantages[edit]
The main advantages of the Fleming–Tamao oxidation include its high selectivity and mild reaction conditions. Unlike other oxidation methods, this reaction does not require harsh conditions or strong acids, making it more compatible with sensitive functional groups. Additionally, the by-products of the reaction are environmentally benign, contributing to the green chemistry credentials of this methodology.
Limitations[edit]
Despite its many advantages, the Fleming–Tamao oxidation has some limitations. The reaction can be sensitive to the steric and electronic properties of the substrate, and in some cases, the desired oxidation may not proceed efficiently. Moreover, the requirement for a silyl protecting group adds an additional step to the synthetic sequence, which can be a drawback in some applications.
See Also[edit]
References[edit]
<references/>
-
Fleming–Tamao oxidation
-
Tamao oxidation mechanism
-
Fleming–Tamao oxidation
-
Phenol generation using FT oxidation
-
Ketone, Aldehyde, Acid
-
Effects of substituents
-
Synthesis of Natural Product
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian