2-Ethoxyethanol
2-Ethoxyethanol[edit]
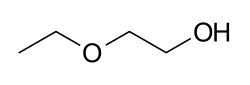
2-Ethoxyethanol, also known as ethylene glycol monoethyl ether, is an organic compound with the formula C4H10O2. It is a clear, colorless liquid with a sweet, ether-like odor. This compound is part of the glycol ether family and is used primarily as a solvent in various industrial applications.
Chemical Properties[edit]
2-Ethoxyethanol is a glycol ether, which means it contains both an ether and an alcohol functional group. Its chemical structure consists of an ethyl group attached to an ethylene glycol moiety. This dual functionality gives it the ability to dissolve both polar and non-polar substances, making it a versatile solvent.
Uses[edit]
2-Ethoxyethanol is used in a variety of applications due to its solvent properties. It is commonly found in:
Its ability to dissolve a wide range of substances makes it valuable in these industries.
Health and Safety[edit]

Exposure to 2-Ethoxyethanol can pose health risks. It is known to be toxic to humans, with potential effects on the reproductive system and developmental toxicity. Prolonged exposure can lead to symptoms such as headache, dizziness, and nausea. It is important to handle this chemical with appropriate safety measures, including the use of personal protective equipment (PPE) and adequate ventilation.
Environmental Impact[edit]
2-Ethoxyethanol can have environmental effects if released into the environment. It is biodegradable, but its presence in water bodies can be harmful to aquatic life. Proper disposal and handling are essential to minimize its environmental impact.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian