Eschweiler–Clarke reaction
Eschweiler–Clarke Reaction
The Eschweiler–Clarke Reaction is a chemical reaction used primarily in organic chemistry for the methylation of primary and secondary amines. This reaction involves the reductive methylation of amines using formaldehyde and formic acid, resulting in the formation of tertiary amines or secondary amines, respectively. The reaction is named after the German chemists Wilhelm Eschweiler and Hans Thacher Clarke, who independently developed the method in the early 20th century.
Reaction Mechanism[edit]
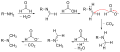
The Eschweiler–Clarke reaction proceeds through a series of steps. Initially, the amine reacts with formaldehyde to form an imine intermediate. Subsequently, this intermediate is reduced by formic acid, leading to the methylation of the nitrogen atom. In the case of primary amines, the process can repeat, allowing for the possibility of double methylation to form tertiary amines. The overall reaction can be summarized as follows:
- For primary amines: R-NH2 + 2 CH2O + 2 HCOOH → R-N(CH3)2 + 3 H2O
- For secondary amines: R2NH + CH2O + HCOOH → R2N-CH3 + 2 H2O
Applications[edit]
The Eschweiler–Clarke reaction is widely used in the synthesis of tertiary amines, which are important intermediates in the production of pharmaceuticals, agrochemicals, and dyes. This reaction is particularly valuable because it offers a straightforward method for the N-methylation of amines, a modification that can significantly alter the biological activity of a compound.
Advantages and Limitations[edit]
One of the main advantages of the Eschweiler–Clarke reaction is its simplicity and the use of readily available and relatively inexpensive reagents. However, the reaction has some limitations, including the potential for over-methylation and the use of formic acid, which can be hazardous in large quantities. Additionally, the reaction conditions may not be suitable for substrates that are sensitive to acidic environments.
Related Reactions[edit]
The Eschweiler–Clarke reaction is related to other reductive alkylation methods, such as the Leuckart Reaction, which also involves the use of formic acid but employs ammonium formate as the reducing agent. Another related reaction is the Bouveault-Blanc Reduction, which is used for the reduction of esters to primary alcohols.
See Also[edit]
References[edit]
- Eschweiler–Clarke reaction
-
Eschweiler–Clarke reaction
-
Eschweiler–Clarke reaction mechanism
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian