Erteberel
Selective estrogen receptor modulator
| Erteberel | |
|---|---|
|
| |
| IUPAC name | (2S)-2-(4-Hydroxyphenyl)-3-methyl-1-[4-(2-pyrrolidin-1-ylethoxy)phenyl]butan-1-one
|
Erteberel (developmental code name LY-500307) is a selective estrogen receptor modulator (SERM) that was under development by Eli Lilly and Company for the treatment of benign prostatic hyperplasia (BPH) and other conditions. It is a nonsteroidal compound that acts as a selective agonist of the estrogen receptor beta (ERβ), with high selectivity over the estrogen receptor alpha (ERα).
Mechanism of Action[edit]
Erteberel functions by selectively binding to and activating the estrogen receptor beta (ERβ). This receptor is one of the two main types of estrogen receptors, the other being estrogen receptor alpha (ERα). The selective activation of ERβ is thought to provide therapeutic benefits in certain conditions without the side effects associated with ERα activation. This selectivity is particularly important in tissues where ERβ is predominantly expressed, such as the prostate gland.
Potential Therapeutic Uses[edit]
Erteberel was primarily investigated for its potential use in treating benign prostatic hyperplasia (BPH), a condition characterized by the enlargement of the prostate gland. By selectively targeting ERβ, erteberel was hypothesized to reduce prostate size and alleviate symptoms associated with BPH without affecting other tissues where ERα is more prevalent.
Other Investigated Uses[edit]
In addition to BPH, erteberel has been studied for its potential effects on other conditions, such as:
- Osteoporosis: Due to its estrogenic effects on bone tissue, erteberel may help in maintaining bone density.
- Cognitive disorders: There is interest in the role of ERβ in the brain, suggesting potential benefits in cognitive health.
Development and Research[edit]
Erteberel was developed by Eli Lilly and Company, a major pharmaceutical company. Despite its promising mechanism of action, the development of erteberel was eventually discontinued. The reasons for discontinuation are not publicly detailed, but it is common for drug development to be halted due to various factors such as efficacy, safety, or strategic business decisions.
Chemical Structure[edit]
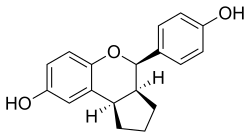
Erteberel is a nonsteroidal compound with a complex chemical structure that allows it to selectively bind to ERβ. Its chemical name is (2S)-2-(4-Hydroxyphenyl)-3-methyl-1-[4-(2-pyrrolidin-1-ylethoxy)phenyl]butan-1-one.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian