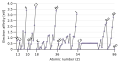Electron affinity
Electron Affinity is a chemical property that quantifies the ability of an atom to accept an electron. It is a measure of the energy change when an electron is added to a neutral atom in the gaseous state to form a negative ion. This property is significant in the study of chemical bonding, ionization energies, and the periodic trends across the Periodic Table.
Definition[edit]
Electron affinity is defined as the amount of energy released or spent when an electron is added to a neutral atom or molecule in the gas phase to form a negative ion. It is usually expressed in units of kilojoules per mole (kJ/mol) or electronvolts (eV).
Trends in the Periodic Table[edit]
Electron affinity varies across the Periodic Table. Generally, it increases across a period from left to right and decreases down a group. However, there are exceptions to this trend. For instance, the electron affinity of elements in Group 17 (the halogens) is particularly high due to their one electron shortfall from achieving a stable noble gas electron configuration.
Factors Affecting Electron Affinity[edit]
Several factors influence the electron affinity of an element:
- Atomic Size: Smaller atoms tend to have higher electron affinity because the added electron is closer to the nucleus, experiencing a stronger attraction.
- Nuclear Charge: A higher effective nuclear charge increases electron affinity as the increased charge exerts a stronger pull on the added electron.
- Electron Configuration: Atoms with a nearly full or half-full valence shell tend to have higher electron affinity because the added electron would lead to a more stable configuration.
Measurement[edit]
Electron affinity can be difficult to measure directly. It is often determined indirectly through various experimental techniques, such as photoelectron spectroscopy.
Applications and Significance[edit]
Understanding electron affinity is crucial in fields such as quantum chemistry, material science, and electrochemistry. It helps in predicting the reactivity of elements, the stability of ions, and the formation of compounds. Electron affinity is also essential in the study of semiconductors and other materials with electronic applications.
Examples[edit]
- The halogens have some of the highest electron affinities because they are one electron away from having a full valence shell.
- Noble gases have low electron affinities because they already possess a complete valence shell, making them stable and less likely to accept an electron.
See Also[edit]
-
Electron affinity of the elements
-
Semiconductor vacuum junction
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian